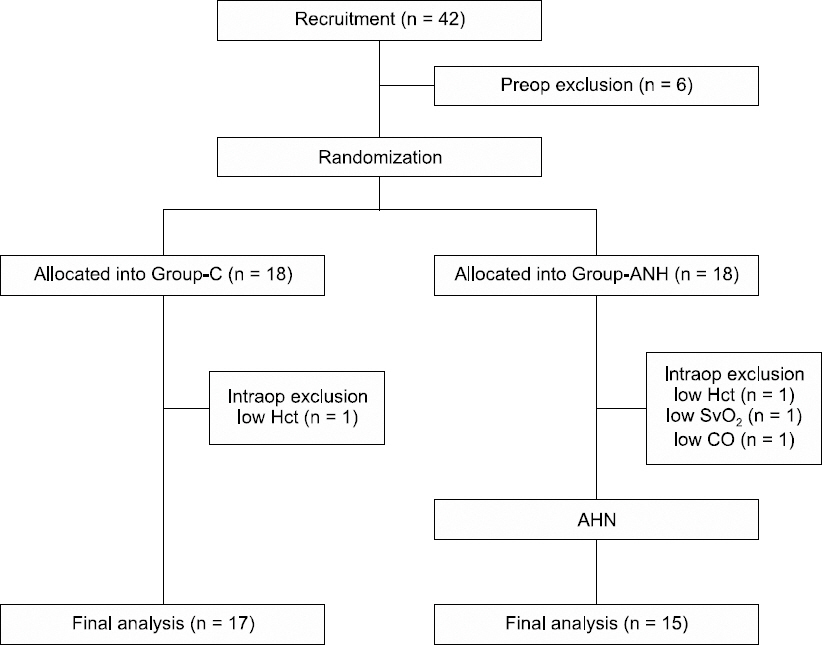
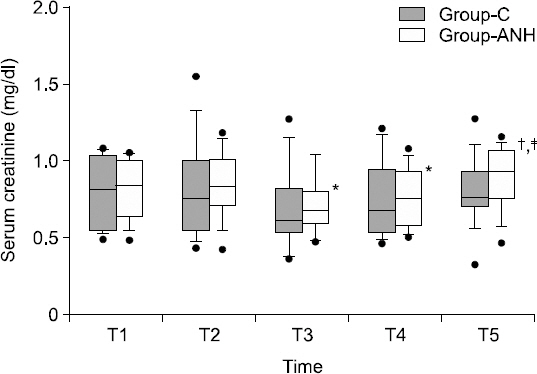
1. Bennett SR. Perioperative autologous blood transfusion in elective total hip prosthesis operations. Ann R Coll Surg Engl 1994; 76: 95-8. PMID:
10.1097/00132586-199412000-00057.



2. Bennett J, Haynes S, Torella F, Grainger H, McCollum C. Acute normovolemic hemodilution in moderate blood loss surgery: a randomized controlled trial. Transfusion 2006; 46: 1097-103. PMID:
10.1111/j.1537-2995.2006.00857.x. PMID:
16836555.


3. Berg KS, Stenseth R, Wahba A, Pleym H, Videm V. How can we best predict acute kidney injury following cardiac surgery?: a prospective observational study. Eur J Anaesthesiol 2013; 30: 704-12. PMID:
10.1097/EJA.0b013e328365ae64. PMID:
24067536.


4. Mangano CM, Diamondstone LS, Ramsay JG, Aggarwal A, Herskowitz A, Mangano DT. Renal dysfunction after myocardial revascularization: risk factors, adverse outcomes, and hospital resource utilization. The Multicenter Study of Perioperative Ischemia Research Group. Ann Intern Med 1998; 128: 194-203. PMID:
10.7326/0003-4819-128-3-199802010-00005. PMID:
9454527.


5. Jung HS, Kim CS, Kim TY. The relationship of serum creatinine and cardiac troponin I after off-pump coronary artery bypass graft surgery. Anesth Pain Med 2009; 4: 124-8.
7. Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL. Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 2007; 71: 1028-35. PMID:
10.1038/sj.ki.5002231. PMID:
17396113.


8. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 2004; 8: R204-12. PMID:
10.1186/cc2671. PMID:
10.1186/cc2872. PMID:
15312219. PMID:
PMC522841.



9. Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, et al. Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 2007; 11: R31PMID:
10.1186/cc5713. PMID:
17331245. PMID:
PMC2206446.



10. Parolari A, Pesce LL, Pacini D, Mazzanti V, Salis S, Sciacovelli C, et al. Risk factors for perioperative acute kidney injury after adult cardiac surgery: role of perioperative management. Ann Thorac Surg 2012; 93: 584-91. PMID:
10.1016/j.athoracsur.2011.09.073. PMID:
22269725.


11. Kumar AB, Suneja M, Bayman EO, Weide GD, Tarasi M. Association between postoperative acute kidney injury and duration of cardiopulmonary bypass: a meta-analysis. J Cardiothorac Vasc Anesth 2012; 26: 64-9. PMID:
10.1053/j.jvca.2011.07.007. PMID:
21924633.


12. Rosner MH, Okusa MD. Acute kidney injury associated with cardiac surgery. Clin J Am Soc Nephrol 2006; 1: 19-32. PMID:
10.2215/CJN.00240605. PMID:
17699187.


13. Bojan M, Lopez-Lopez V, Pouard P, Falissard B, Journois D. Limitations of early serum creatinine variations for the assessment of kidney injury in neonates and infants with cardiac surgery. PLoS One 2013; 8: e79308PMID:
10.1371/journal.pone.0079308. PMID:
24244476. PMID:
PMC3823616.



14. Waikar SS, Liu KD, Chertow GM. Diagnosis, epidemiology and outcomes of acute kidney injury. Clin J Am Soc Nephrol 2008; 3: 844-61. PMID:
10.2215/CJN.05191107. PMID:
18337550.


15. Lassnigg A, Schmid ER, Hiesmayr M, Falk C, Druml W, Bauer P, et al. Impact of minimal increases in serum creatinine on outcome in patients after cardiothoracic surgery: do we have to revise current definitions of acute renal failure? Crit Care Med 2008; 36: 1129-37. PMID:
10.1097/CCM.0b013e318169181a. PMID:
18379238.


16. Lassnigg A, Schmidlin D, Mouhieddine M, Bachmann LM, Druml W, Bauer P, et al. Minimal changes of serum creatinine predict prognosis in patients after cardiothoracic surgery: a prospective cohort study. J Am Soc Nephrol 2004; 15: 1597-605. PMID:
10.1097/01.ASN.0000130340.93930.DD. PMID:
15153571.


17. Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW. Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol 2005; 16: 3365-70. PMID:
10.1681/ASN.2004090740. PMID:
16177006.


18. Swaminathan M, Phillips-Bute BG, Conlon PJ, Smith PK, Newman MF, Stafford-Smith M. The association of lowest hematocrit during cardiopulmonary bypass with acute renal injury after coronary artery bypass surgery. Ann Thorac Surg 2003; 76: 784-91. PMID:
10.1016/S0003-4975(03)00558-7.


20. Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16: 31-41. PMID:
10.1159/000180580.


21. Edwards KD, Whyte HM. Plasma creatinine level and creatinine clearance as tests of renal function. Australas Ann Med 1959; 8: 218-24. PMID:
13819423.


22. Stafford-Smith M. Perioperative renal dysfunction: Implications and strategies for protection. In: Perioperative Organ Protection Newman MF. In: Richmond, Society of Cardiovascular Anesthesiologists 2003; 89-124.