|
 |
- Search
| Anesth Pain Med > Volume 18(4); 2023 > Article |
|
Abstract
Background
Endobronchial ultrasound (EBUS) is widely used to diagnose lung cancer. Monitored anesthesia care (MAC) can enhance patient comfort and procedural conditions during EBUS. EBUS under MAC is usually safe but can lead to various complications.
Case
A 34-year-old male who had increased sputum for two months showed an enlarged paratracheal lymph node and planned for lymph node biopsy by EBUS. During EBUS under MAC, an unexpected oxygen saturation decline required intervention. After intubation, copious frothy fluid was suctioned from the bronchi, and oxygenation was recovered. A narrowed trachea and the EBUS bronchoscope might have resulted in upper airway obstruction, and suction performed under these conditions might have caused pulmonary edema. The patient received non-invasive ventilation and high-flow nasal cannula and recovered without complications.
Integrating endoscopy and ultrasound in endobronchial ultrasound (EBUS) enhances the depiction of mediastinal structures and facilitates the observation of parabronchial anatomy. This results in a more accurate selection of sites for transbronchial sampling and reduces biopsy sampling errors [1]. The outer diameter of EBUS (6.3-6.9 mm) is larger than that of a conventional bronchoscope (4.9-6.0 mm). EBUS with transbronchial needle aspiration (TBNA) is a commonly used technique for diagnosing, staging, and restaging suspected or confirmed non-small cell lung cancer cases. TBNA is also used for sampling mediastinal lymphadenopathy, masses, pulmonary nodules, and endobronchial lesions. Compared with mediastinoscopy or percutaneous needle biopsy, EBUS-TBNA is less invasive, making it an advantageous option [2].
During the EBUS-TBNA procedure, sedatives are administered because a thicker scope is used than a standard bronchoscope, has a longer procedure time, and biopsy can be challenging because of patient movement. The type of sedation can vary from minimal sedation to general anesthesia, depending on the institution [3,4]. EBUS-TBNA is typically performed under monitored anesthesia care (MAC) at our hospital. This report describes a case of unexpected severe pulmonary edema during EBUS-TBNA for paratracheal lymph node biopsy under MAC.
Written informed consent was obtained to publish this case report from the patient. The Institutional Review Board waived the review of this case report (IRB No. 2023-03-017).
A 34-year-old male (169.4 cm, 66.7 kg) presented to the hospital with persistent sputum for 2 months. He had no specific underlying disease and was not taking any medications. Chest computed tomography (CT) revealed multiple enlarged nodes in the mediastinum. We planned to perform EBUS under MAC, targeting moderate sedation that allows spontaneous ventilation in conjunction with a high-flow nasal cannula (HFNC). Before the procedure, the patient’s vital signs were stable, with a blood pressure of 119/83 mmHg, heart rate of 88 beats per min, respiratory rate of 20 breaths per min, oxygen saturation (SpO2) of 98%, and body temperature of 36.3°C. During the procedure, HFNC (Optiflow THRIVE, Fisher & Paykel Healthcare) was used to supply oxygen with a fraction of inspired oxygen (FiO2) of 1.0 and a flow of 40 L/min. Sedation was achieved with a bolus of remimazolam 5 mg and a target-controlled infusion of remifentanil at a target site concentration of 2.0 ng/ml. Before the procedure started, the patient’s sedation level was confirmed to be moderate. During the procedure, remimazolam was administered in bolus doses of 2.5 mg as needed (a total of 20 mg used) to maintain sedation depth, and remifentanil was adjusted to a range of 2.0-3.0 ng/ml.
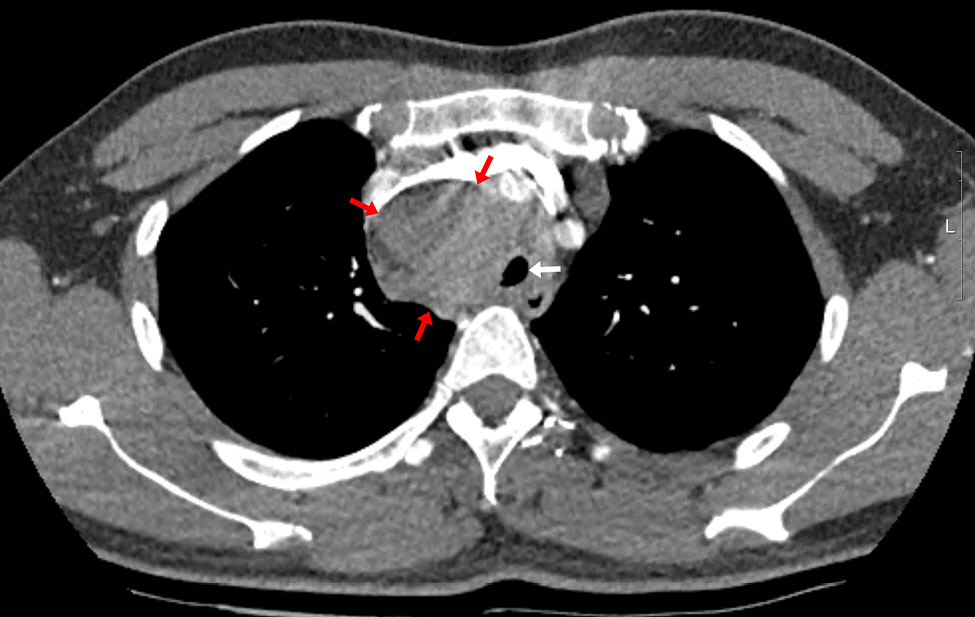
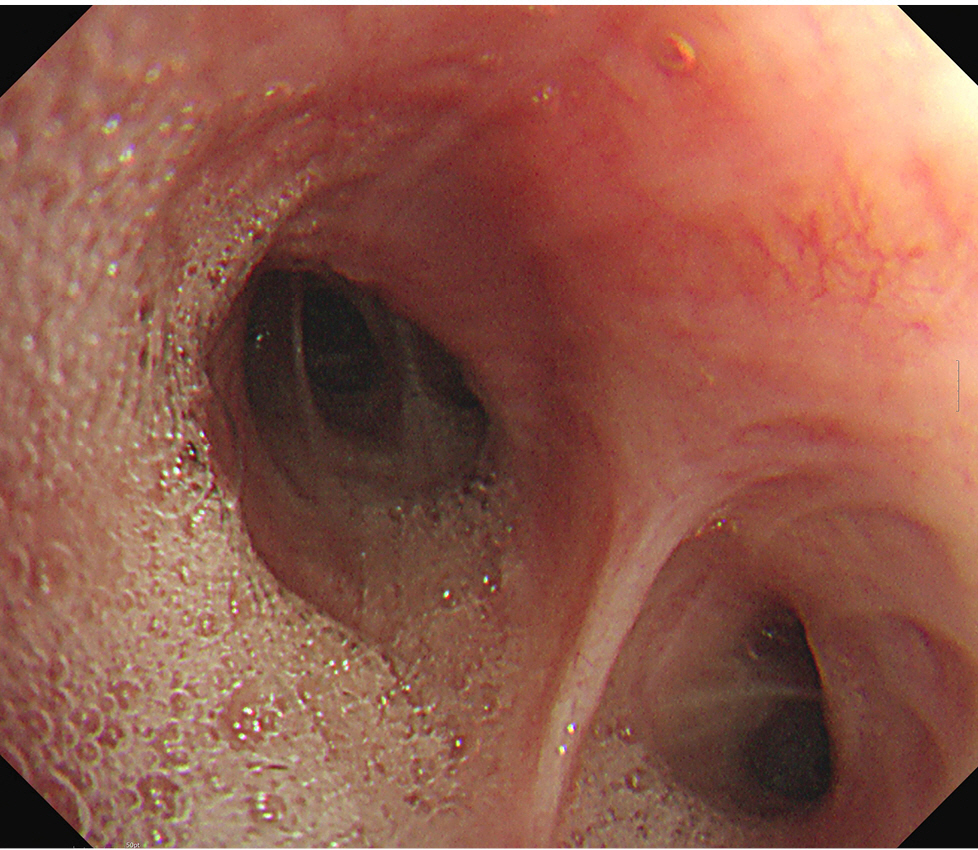
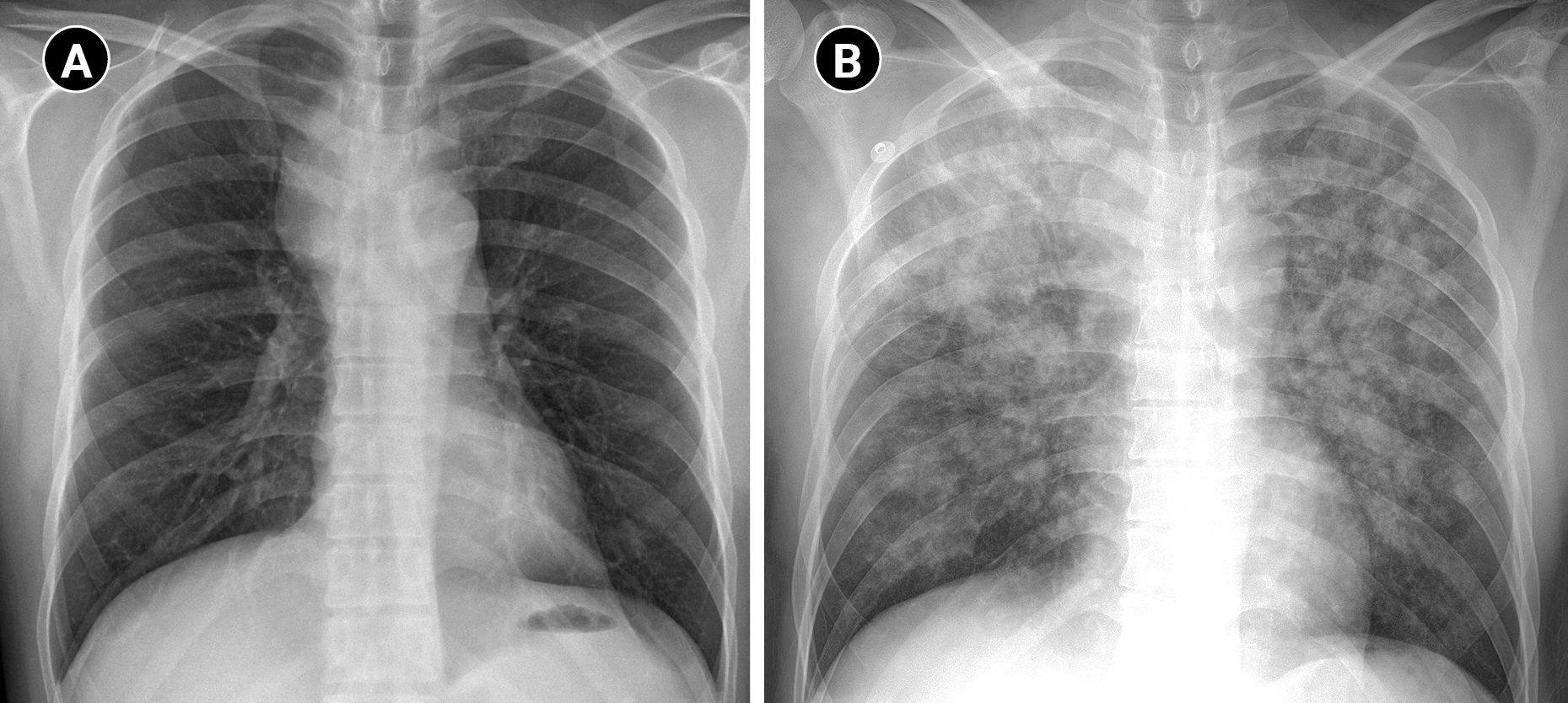
The patient had an enlarged lymph node (4.9 × 4.1 cm) in the right paratracheal area on chest CT, resulting in tracheal narrowing (7.3 mm) (Fig. 1). No problems were encountered while inserting the flexible bronchoscope (BF-H290, Olympus), which had a 5.7 mm diameter, and no specific abnormalities were found in the entire bronchus. During the subsequent EBUS (BF-UC260FW, Olympus) insertion with a 6.9 mm diameter, mild resistance was encountered. The procedure started with bronchial suctioning for secretion removal to ensure a clear visual field. About 3 min after the insertion of the EBUS, the patient’s spontaneous breathing was maintained, but SpO2 began to decrease below 90%. The procedure was stopped, and we applied a jaw thrust maneuver. However, as the saturation decreased, the EBUS bronchoscope was removed, and a jaw thrust was continued. However, the patient exhibited weak self-respiration, and SpO2 rapidly decreased to approximately 30%. Bag-mask ventilation was immediately started, but the inspiratory pressure was high, and end-tidal CO2 (EtCO2) was also inconsistent, indicating inefficient ventilation. Within 1-2 min, SpO2 slowly increased to 90%, but it did not increase further despite FiO2 of 1.0. No secretions were observed on oral suction. We planned to reinsert the flexible bronchoscope to evaluate the cause of hypoxia. However, because SpO2 was maintained at approximately 90% with positive-pressure mask ventilation, we thought it would be difficult to advance the bronchoscope with MAC, which has an insecure airway and depends on self-respiration. Therefore, we decided to proceed with bronchoscopy under mechanical ventilation after intubation. Because the biopsy site was not easily accessible after endotracheal tube (ETT) insertion owing to its location close to the vocal cord, the operator asked for a supraglottic airway device. However, the authors decided to use an ETT, considering the patient might require high positive airway pressure. To facilitate intubation, we administered an additional 50 mg of propofol and 40 mg of succinylcholine. After successful intubation with an ETT with an internal diameter of 7.5 mm, a large amount of pink frothy fluid was regurgitated through the ETT, and SpO2 was restored to 95% after tracheal suction. Although continuous secretion was regurgitated, SpO2 was maintained at approximately 90-95% with FiO2 of 1.0. A bronchoscopic exam revealed a large amount of frothy fluid throughout the bronchi (Fig. 2), and sufficient suction was performed. The patient could maintain SpO2 without high positive-pressure ventilation after sufficient suction during bronchoscopy. The peak inspiratory pressure was reduced from 26 to 20 mmHg after bronchoscopic toileting of the secretion, at a tidal volume of 450 ml, respiratory rate of 14/min, and positive end-expiratory pressure (PEEP) of 5 cmH2O. Subsequently, for the paratracheal lymph node biopsy, we administered propofol 20 mg and succinylcholine 30 mg, followed by the replacement of the ETT with a size 4 supraglottic airway device (i-gel, Intersurgical Ltd.). Biopsy using an EBUS bronchoscope was performed while maintaining positive-pressure ventilation through a supraglottic airway device. SpO2 was maintained around 90% with FiO2 of 1.0 for the duration of the procedure. After the procedure, the patient was extubated, and HFNC with FiO2 of 1.0 and flow rate of 40 L was applied, maintaining SpO2 above 90%. In the post-anesthesia care unit, the patient coughed severely and expectorated pink frothy sputum in a sitting position, but SpO2 was maintained at approximately 95%. We gradually reduced the FiO2 and flow rate to 0.75 and 30 L in 30 min while maintaining SpO2 of 95%. The patient was transferred to the intensive care unit. Chest radiography after transfer showed bilateral pulmonary edema (Fig. 3).
Overnight non-invasive ventilation was applied with inspiratory positive airway pressure of 11 cmH2O (above PEEP: 5 cmH2O), expiratory positive airway pressure of 6 cmH2O and FiO2 of 0.4 on the day of the procedure, and HFNC (FiO2 of 0.4 and a flow rate of 40 L/min) was maintained on postoperative day (POD) 1. On POD 2, the patient's SpO2 was above 90% using HFNC, so the patient was transferred to the general ward. The patient’s SpO2 was maintained at 98% in the general ward using HFNC and subsequently switched to nasal prongs with an oxygen flow rate of 3 L. The target SpO2 was 94%, and O2 was successfully tapered up to 1 L of oxygen. On POD 3, the patient's SpO2 was above 95% without supplemental O2. With no additional issues, the patient was discharged on POD 5.
EBUS is generally considered a safe procedure but may lead to various complications. Complications can be broadly classified into two categories: procedure-related and sedation-related. Procedure-related complications include agitation, cough, blood at the puncture site, pneumothorax, bronchospasm, and laryngospasm. Moderate bleeding has been reported in 1% of cases, and pneumothorax has been reported in less than 4% [5]. Sedation-related complications include respiratory depression with hypoxia or hypercarbia, cardiovascular instability, and inadequate sedation [6]. These complications can frequently result in desaturation either alone or in combination.
The primary cause of severe acute desaturation in this case was pulmonary edema, rather than commonly reported causes. Despite thorough suctioning via the ETT and the need for extensive bronchoscope-assisted suctioning, pink frothy sputum persisted in the recovery room, indicating severe pulmonary edema. In patients with upper airway obstruction, negative pressure pulmonary edema (NPPE) can occur when accompanied by high negative intrathoracic pressure (NIP) [7]. NPPE occurs more frequently in healthy young individuals who can generate large NIP, and up to 12% of NPPE cases occur after upper airway obstruction [8]. This patient had a tracheal narrowing due to a paratracheal lymph node. Moreover, considering the diameter of the EBUS bronchoscope and the resistance encountered during insertion, it might have sealed the trachea and made an upper airway obstruction. In such a situation, spontaneous ventilation with high NIP can potentially contribute to NPPE and cannot be completely ruled out. However, we administered MAC with sufficient sedatives and opioids that suppressed cough and excessive ventilatory drive. No excessive ventilation or significant paradoxical movements of the thorax or abdomen that could generate negative pressure until the desaturation event was observed. Therefore, we considered that continuous suctioning during the EBUS procedure in patients with narrow or obstructed upper airways could generate high, continuous NIP, which might be a possible cause of NPPE. In adults, suction pressure is recommended to be below negative 200 mmHg [9]. The suction used during EBUS had a maximal negative pressure of 500 mmHg, meaning that approximately 200 mmHg would be applied in an open cavity. While this may not be a problem in an open cavity, it could lead to high NIP if there is upper airway obstruction. One case similar to ours has been reported, in which pulmonary edema occurred due to suction during bronchoscopy in an infant [10].
There are other factors that can lead to upper airway obstruction, such as laryngospasm and bronchospasm. This could potentially be a cause of desaturation and may also serve as another potential contributor to NPPE. Stimulation of the larynx and bronchus by the bronchoscope, especially with inadequate sedation, can cause airway irritation, leading to spasms [11]. In this case, the high peak inspiratory pressure and the inaccurate capnography waveform during the initial bag-mask ventilation after the desaturation event may suggest the possibility of airway spasm. This could be one of the factors contributing to the initial desaturation and may also be another factor in upper airway obstruction leading to NPPE.
MAC itself can also cause upper airway obstruction and desaturation. Respiratory depression with hypoxia or hypercarbia is a common side effect when sedatives and opioids are used together during the procedure [12]. In our case, we maintained an adequate sedation level, preserving spontaneous breathing, and the initial SpO2 was well maintained when a flexible bronchoscope was used during the procedure before EBUS. However, it is difficult to completely exclude the effect of a compromised airway by MAC.
While it is unlikely to be the cause in our case, the following reasons could potentially contribute to desaturation during EBUS. Prolonged suctioning during bronchoscopy can cause further de-recruitment and atelectasis, leading to post-procedural hypoxemia [13]. Bleeding can cause desaturation. During the bronchoscopic biopsy, a small amount of bleeding can occur at the puncture site. Almost all bleeding events are resolved with conservative treatment [14]. In some cases, such blood and mucus can create atelectasis and airway obstruction, leading to desaturation, which may require bronchial washing. Pneumothorax can also cause desaturation [15].
Performing EBUS-TBNA under MAC has several advantages. An anesthesiologist can perform a preoperative airway evaluation and consult with the operator in advance. Proper selection and dose adjustment of sedatives and analgesics by an anesthesiologist can reduce hemodynamic instability and aid in airway reflex suppression. Most importantly, the presence of an anesthesiologist during the procedure allows for quick intervention and accurate airway management in an unexpected event. However, even with skilled anesthesiologists present, efforts such as using a nasal airway, monitoring sedation depth with tools like bispectral index, and confirming ventilation with end-tidal CO2 may be necessary for early detection of complications and precise drug administration. Furthermore, prior to any bronchoscopic procedure where airway stability cannot be guaranteed, a more detailed pre-anesthetic evaluation, including airway, trachea, and lung condition assessment, should be conducted to anticipate potential issues.
In conclusion, when performing EBUS-TBNA under MAC, various causes of desaturation events can occur. Unexpected pulmonary edema, as observed in this case, can occur, especially when there are anatomical issues in the upper airway. Anesthesiologists should focus on more detailed preoperative evaluations and vigilant monitoring to prevent complications.
Notes
CONFLICTS OF INTEREST
Jun Hyun Kim is the current deputy editor, of Anesthesia and Pain Medicine. However, he was not involved in the peer reviewer selection, evaluation, or decision process of this article.
DATA AVAILABILITY STATEMENT
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
AUTHOR CONTRIBUTIONS
Conceptualization: Kyung Woo Kim, Jun Hyun Kim, Ji Yeon Kim. Writing-original draft: Hwan Yong Choi, Kyung Woo Kim. Writing-review & editing: Hyung Koo Kang, Min Hee Heo, Kyung Woo Kim, Jun Hyun Kim, Sang Il Lee, Ji Yeon Kim, Kyung-Tae Kim, Jang Su Park, Won Joo Choe. Supervision: Kyung Woo Kim, Jun Hyun Kim.
REFERENCES
1. Jiang J, Browning R, Lechtzin N, Huang J, Terry P, Wang KP. TBNA with and without EBUS: a comparative efficacy study for the diagnosis and staging of lung cancer. J Thorac Dis 2014; 6: 416-20.


2. Um SW, Kim HK, Jung SH, Han J, Lee KJ, Park HY, et al. Endobronchial ultrasound versus mediastinoscopy for mediastinal nodal staging of non-small-cell lung cancer. J Thorac Oncol 2015; 10: 331-7.


3. Casal RF, Lazarus DR, Kuhl K, Nogueras-González G, Perusich S, Green LK, et al. Randomized trial of endobronchial ultrasound-guided transbronchial needle aspiration under general anesthesia versus moderate sedation. Am J Respir Crit Care Med 2015; 191: 796-803.



4. Douglas N, Ng I, Nazeem F, Lee K, Mezzavia P, Krieser R, et al. A randomised controlled trial comparing high-flow nasal oxygen with standard management for conscious sedation during bronchoscopy. Anaesthesia 2018; 73: 169-76.



5. Hayama M, Izumo T, Matsumoto Y, Chavez C, Tsuchida T, Sasada S. Complications with endobronchial ultrasound with a guide sheath for the diagnosis of peripheral pulmonary lesions. Respiration 2015; 90: 129-35.



6. Miller MA, Levy P, Patel MM. Procedural sedation and analgesia in the emergency department: what are the risks? Emerg Med Clin North Am 2005; 23: 551-72.


7. Lee JH, Lee JH, Lee MH, Cho HO, Park SE. Postoperative negative pressure pulmonary edema following repetitive laryngospasm even after reversal of neuromuscular blockade by sugammadex: a case report. Korean J Anesthesiol 2017; 70: 95-9.




8. Tami TA, Chu F, Wildes TO, Kaplan M. Pulmonary edema and acute upper airway obstruction. Laryngoscope 1986; 96: 506-9.


9. Blakeman TC, Scott JB, Yoder MA, Capellari E, Strickland SL. AARC clinical practice guidelines: artificial airway suctioning. Respir Care 2022; 67: 258-71.


10. Hannania S, Barak M, Katz Y. Unilateral negative-pressure pulmonary edema in an infant during bronchoscopy. Pediatrics 2004; 113: e501-3.



11. Bedolla-Pulido TR, Bedolla-Barajas M. [Spontaneous pneumomediastinum and subcutaneous emphysema associated with bronchospasm in a woman with no history of asthma]. Rev Alerg Mex 2017 64: 386-9. Spanish.

12. Practice Guidelines for Moderate Procedural Sedation and Analgesia 2018: A Report by the American Society of Anesthesiologists Task Force on Moderate Procedural Sedation and Analgesia, the American Association of Oral and Maxillofacial Surgeons, American College of Radiology, American Dental Association, American Society of Dentist Anesthesiologists, and Society of Interventional Radiology. Anesthesiology 2018; 128: 437-79.

13. Lindholm CE, Ollman B, Snyder JV, Millen EG, Grenvik A. Cardiorespiratory effects of flexible fiberoptic bronchoscopy in critically ill patients. Chest 1978; 74: 362-8.


- ARTICLE & TOPICS
-
- Topics
-
- Neuroscience in anesthesiology and critical care
- Anesthetic Pharmacology
- Obstetric Anesthesia
- Pediatric Anesthesia
- Cardiothoracic and Vascular Anesthesia
- Transplantation Anesthesia
- Spinal Pain
- Regional Anesthesia
- Neuromuscular Physiology and Pharmacology
- Airway Management
- Geriatric anesthesia and Pain
- Others