|
 |
- Search
| Anesth Pain Med > Volume 18(1); 2023 > Article |
|
Abstract
Background
Lymphedema is characterized by localized tissue swelling due to excessive interstitial space retention of lymphatic fluid. Lymphedema is easy to be misdiagnosed since it resembles other conditions of extremity swelling. We present a case of complex regional pain syndrome (CRPS) type I with secondary lymphedema that was successfully managed with spinal cord stimulation (SCS).
Case
A 39-year-old female patient came to our pain clinic with complaints of lower extremity pain and edema. To find out reason of leg edema, computed tomography of extremity angiography and blood test were performed. However, all of evaluations were normal. Lastly performed lymphoscintigraphy showed secondary lymphedema. SCS was performed and it showed dramatic reduction subsequent to implantation of SCS.
Complex regional pain syndrome (CRPS) is known as one of the most severe and disabling condition which significantly impair the quality of life with psychologic distress. The diagnosis and treatment of CRPS are challenging due to the lack of confirmative diagnostic test and definitive treatment tools [1]. The Budapest criteria, which shows greatly improved diagnostic specificity compared to international association of study of pain criteria, is widely used to diagnose CRPS [2]. Earlier detection and an interdisciplinary cooperation for treatment seem to be essential in alleviating the symptoms of CRPS, although one single treatment has not been found to be effective definitely.
Lymphedema is characterized by localized tissue swelling due to excessive interstitial space retention of lymphatic fluid caused by obstructed lymphatic drainage. Primary lymphedema is rare and it is caused by genetic or developmental lymphatic vascular anomalies. Most of lymphedema which we encounter is secondary and this disorder is caused by an underlying carcinoma, parasite infection, trauma, or surgery. Lymphedema is easy to be misdiagnosed frequently since it resembles other conditions of extremity swelling [3].
We present a case of CRPS type I which was combined with secondary lymphedema and successfully managed with spinal cord stimulation (SCS).
The authors certify that written informed consent for publication was obtained from the patient or guardian. The potential risks and benefits were discussed with the patient before the spinal cord stimulation.
A female patient of 39 years old, who complained of severe right side lower extremity pain, visited our pain clinic. Her right side extremity pain started one month ago and her numerical rating scale (NRS) was 7. In addition to severe pain, she felt redness, heat, tenderness, and weakness of right leg during her walking. Mild edema was also present. The color of right leg was more reddish compared to left leg. She had a prior history of fracture of right foot and subsequent cast maintenance for one month. Because of her severe pain, color change, local heat and hypoesthesia, we suspected her CRPS. For the diagnostic work up of CRPS, electromyography (EMG), nerve conduction velocity (NCV), 3-phase bone scan, and quantitative sudomotor axon reflex test (QSART) were performed. The result of EMG and NCV was normal. QSART demonstrated skin temperature asymmetry, with the affected right leg being warmer than the left leg. The greatest temperature difference (2.0°C) was observed in an ankle area. Resting sweat out was normal, but Q-sweat output was greater on right side. The 3-phase bone scan showed increased tracer uptake in delayed skeletal phase. According to the clinical feature and the results of diagnostic work up, we diagnosed her CRPS type I of lower extremity. For her pain relief, medication was started including pregabalin 150 mg/day, oxycodone 20 mg/day, and amitriptyline 10 mg/day. However, those medications showed minimal effect in relieving her pain. Lumbar sympathetic ganglion block was performed 3 times for the improvement of pain and edema and it showed temporary relief of pain.
Two weeks after treatment of CRPS, she started to complain of both side leg pain. At first, only right side was painful, but left side leg became painful gradually. In addition to pain, both legs presented severe edema. Since one of important clinical feature of CRPS is edema, we just observed her leg edema using medication to improve edema without further evaluation. Lumbar sympathetic ganglion block improved her leg pain and edema, but it lasted only 2-3 days. She felt severe heaviness and discomfort of leg. Also, she was very difficult to walk due to severe leg edema.
Although CRPS can present the symptom of edema, we thought that edema of this patient is somewhat different from that of CRPS. As a reason of such suspicion, this edema showed pitting edema, and over time, the skin became indurated with hard and leathery texture. Lower extremity elevation during sleep did not improve the edema. Due to a different nature of leg edema, we suspected venous edema, drug induced swelling, cellulitis, and lymphedema for the possible cause of edema.
To find out possible reason of leg edema, computed tomography (CT) of extremity angiography and laboratory blood test were performed. The result of CT angiography was completely normal. Blood test including C reactive protein and erythrocyte sedimentation rate was also within normal limit except for mildly increased liver enzyme. She was not taking any medications which are known to cause swelling.
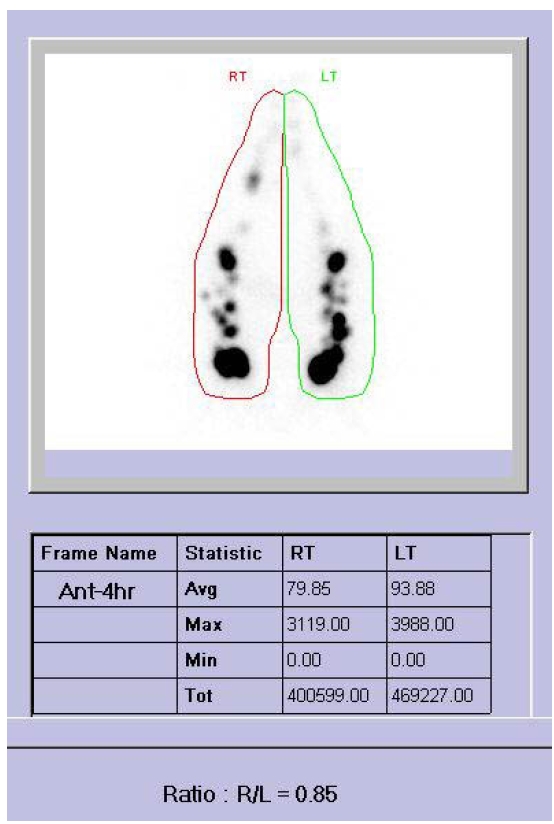
To evaluate lymphedema as a reason of leg edema, she was consulted to rehabilitation department. Both leg girths were measured at 5 regions at proximal 10 cm and 20 cm of patella upper border and distal 10 cm, 20 cm, and 30 cm of patella lower border to assess the severity of edema. The edema was more severe in right side and upper thigh than left side and lower calf. Lastly, lymphoscintigraphy was performed and it showed secondary lymphedema of both lower extremities (Fig. 1). We assessed her both leg CRPS type I combined with lymphedema.
Since her leg edema and pain were still intractable in opioid medication and lumbar sympathetic ganglion block, SCS was performed subsequent to an admission to hospital.
After local infiltration of skin with 1% lidocaine, a 14-gauge needle was inserted into the L2-3 interlaminar space using fluoroscopic guidance. When loss of resistance was felt using distilled water, a steerable guide wire was inserted to confirm the epidural space. When the position of guide wire was in posterior epidural space by fluoroscopy, bilateral octapolar lead was inserted and advanced up to superior endplate of T11, which covers posterior epidural space from T12 to T11 (Fig. 2).
After placement of electrode into the epidural space, tonic mode SCS test stimulation was performed with pulse frequencies of 40-60 Hz, pulse width of 200-500 μs and an intensity of 1.7-2.7 mA at the operating room. After this test stimulation, she was sent to an admission room to observe the proper electrical stimulation for 5 days. The first day of test stimulation period, she was lying in bed all day long to minimize the lead migration and the intensity and the location of electrical stimulation were similar to that of test stimulation at operating room. Sitting was permitted beginning on the third day of test stimulation. However, the stimulation was relatively weak when she was in sitting position compared to lie in bed. The pulse frequencies and pulse width were similar with that of settings at the operating room but the intensity was maintained with 2.5 mA. With such intensity, she could not feel any weakness and differences between sitting and lying position were minimal. We made a programming so that a patient could feel maximal electrical sensation at both upper thigh which corresponds to the most painful site. During 5 days of trial period of SCS, her were reduced to NRS 1-2. After confirming successful reduction of pain and edema, left lower subcutaneous abdominal pocket was made to insert implantable pulse generator (IntellisTM, Medtronic, USA) for permanent SCS. Following permanent SCS, we set the pulse frequency to 50 Hz, the pulse width to 450 μs, and the intensity to 2.5 mA.
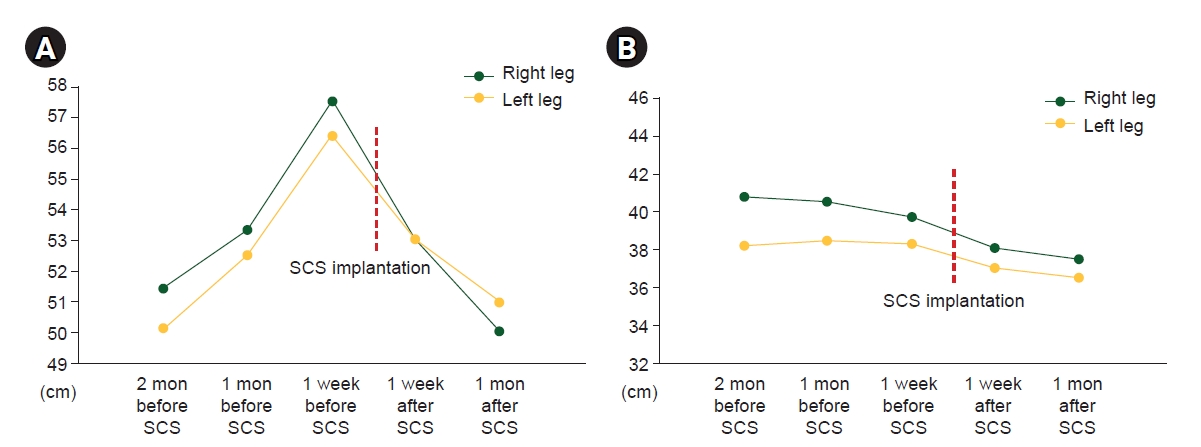
For the evaluation of edema, she was consulted to the rehabilitation department and both leg girths were measured at 5 regions again. Among the 5 regions of measured leg girth, dramatic reduction of edema was observed at upper thigh (proximal 20 cm of patella upper border) and calf (distal 10 cm of patella lower border). Therefore, serial changes of leg girth at those two regions before and after SCS were presented (Fig. 3A, B). In addition to reduction of leg girth, her fibrotic and leathery texture became normalized and disappeared leg hair due to leathery and hard skin started to grow again.
Two months after SCS, NRS was maintained within 1-2 during rest in bed, but NRS increased slightly when she walked around.
This case report demonstrated dramatic improvement of lymphedema using SCS which was combined in CRPS.
Patients diagnosed with CRPS present their symptoms after minor or moderate trauma or tissue injury. The injured extremity shows extremely painful, red, warm, and swollen during the acute phase. Other features which are observed in CRPS include allodynia, hyperalgesia, changes in sweating, hair, nail growth, and muscle weakness. As the disorder persists, pain does not subside but often spreads proximally and can even emerge on the contralateral extremity [4] like a patient of this case report.
If a patient presents severe edema which results from other disease, it is not easy to suspect other reasons because edema in an affected extremity of CRPS is commonly found during their clinical course [4]. At first, we also did not suspect other disease as a cause of edema and just observed it without any further evaluation. However, as the day progressed, her edema became worsen. Moreover, edema of this patient was different from that of CRPS. There are little clinical studies showing specific characteristic of edema which is usually observed in CRPS. It is known that the skin of edematous extremity of CRPS demonstrates a glossy pattern [4]. However, if an edema develops due to lymphedema, the skin becomes hard and fibrotic, and presents even leathery texture with loss of hair [3]. The reason of suspicion that this patient might have other disease was its severity and different nature of edema described above. However, if an edema is combined with trophic change of CRPS, we think that it is hard to differentiate it from lymphedema. Generally, trophic change is usually found in later stage of CRPS, which presents an edema infrequently [1].
Lymphedema is more commonly found in females than in males. Also, lower extremity lymphedema is more frequent than that of upper extremity. Patients diagnosed with lymphedema mostly have a secondary cause. It is reported that the incidence of secondary lymphedema is 1 in 1000 individuals, with the mean age at the time of diagnosis between 50 to 58 years old [3]. Lower extremity lymphedema is usually associated with filariasis (parasite infection), chronic venous insufficiency, obesity, rapamycin treatment in patients with decreased renal function, and malignancies such as lymphoma, uterine cancer, and melanoma. Among causes of secondary lymphedema, parasite infection caused by mosquito-borne nematode is most common. It infects patients who have traveled to endemic area with this disease, usually in India and sub-Saharan Africa [3,5]. However, she did not travel any endemic area, nor have been diagnosed with malignancy. Also, she did not have a history of rapamycin treatment. The obesity was suspected for the reason of development of secondary lymphedema, since her body mass index was 28 kg/m2.
For many years, the primary tool to confirm the presence of lymphedema was lymphoscintigraphy. It visualizes the functional status of the lymphatic system and helps to differentiate between partial and complete obstruction of lymphatic system. Moreover, it can guide a further strategy plan of treatment [3,6]. The advantage of this imaging method is that it is a noninvasive technique which is available at most hospitals. Also, it is easy to perform involving a subcutaneous injection of particulate radiotracer attached to technetium-99m in the distal aspect of the swollen extremity with subsequent imaging of the lymphatic vessel [3,6].
Widely accepted conservative treatment for lymphedema is complex decongestive therapy, which means manual lymphatic drainage massage [7]. Although there is no study suggesting effective management of lymphedema using SCS, this patient experienced effective pain and edema reduction. The exact mechanism of action why the extremity edema caused by lymphedema is improved by SCS is uncertain. The composition of lymphatic system is tonsil, spleen, lymph nodes, and the thymus, all of which are interconnected via a network of lymphatic vessels that run parallel to the venous circulation. Most of interstitial fluid (> 90%) which leaks from capillaries into the tissue is reabsorbed via the venous microcirculation and returns to the blood stream. Remaining interstitial fluid (< 10%) has a high protein concentration and this interstitial fluid (lymph) in drained in blind-ended lymphatic capillaries [3,8]. Once the protein rich interstitial fluid comes into the lymphatic capillaries, it ultimately reenters the circulatory system via collecting lymphatic vessels. In contrast to lymphatic capillaries, collecting lymphatic vessel have smooth muscle walls which has the potential to contract and propel the lymphatic fluid forward [3]. It is assumed that SCS might affect the smooth muscle of collecting lymphatic vessel, ultimately resulting in increasing the ability of lymphatic fluid drainage. Similarly, the restoration and improvement of pulsatile blood flow to the distal extremity in patients of chronic limb ischemia has been reported previously [9-11].
In conclusion, we could successfully manage the intractable pain and edema in patients of CRPS combined with lymphedema. If a patient presents different nature of edema, we need to consider the possibility of coexistence of other disease.
Notes
REFERENCES
1. Kessler A, Yoo M, Calisoff R. Complex regional pain syndrome: an updated comprehensive review. NeuroRehabilitation 2020; 47: 253-64.


2. Harden NR, Bruehl S, Perez RSGM, Birklein F, Marinus J, Maihofner C, et al. Validation of proposed diagnostic criteria (the "Budapest criteria") for complex regional pain syndrome. Pain 2010; 150: 268-74.



3. Grada AA, Phillips TJ. Lymphedema: pathophysiology and clinical manifestations. J Am Acad Dermatol 2017; 77: 1009-20.

4. Marinus J, Moseley GL, Birklein F, Baron R, Maihöfner C, Kingery WS, et al. Clinical features and pathophysiology of complex regional pain syndrome. Lancet Neurol 2011; 10: 637-48.



6. Pappalardo M, Cheng MH. Lymphoscintigraphy for the diagnosis of extremity lymphedema: current controversies regarding protocol, interpretation, and clinical application. J Surg Oncol 2020; 121: 37-47.



7. Thompson B, Gaitatzis K, Janse de Jonge X, Blackwell R, Koelmeyer LA. Manual lymphatic drainage treatment for lymphedema: a systematic review of the literature. J Cancer Surviv 2021; 15: 244-58.



9. Naoum JJ, Arbid EJ. Spinal cord stimulation for chronic limb ischemia. Methodist Debakey Cardiovasc J 2013; 9: 99-102.



-
METRICS

-
- 0 Crossref
- 1,821 View
- 86 Download
- Related articles in Anesth Pain Med
-
An anesthetic experience in a patient with Stickler sydrome: A case report2011 January;6(1)
- ARTICLE & TOPICS
-
- Topics
-
- Neuroscience in anesthesiology and critical care
- Anesthetic Pharmacology
- Obstetric Anesthesia
- Pediatric Anesthesia
- Cardiothoracic and Vascular Anesthesia
- Transplantation Anesthesia
- Spinal Pain
- Regional Anesthesia
- Neuromuscular Physiology and Pharmacology
- Airway Management
- Geriatric anesthesia and Pain
- Others