Choice of the correct size of endotracheal tube in pediatric patients
Article information
Abstract
Selection and insertion of an endotracheal tube (ETT) of appropriate size for airway management during general anesthesia in pediatric patients is very important. A very small ETT increases the risk of inadequate ventilation, air leakage, and aspiration, whereas a very large ETT may cause serious complications including airway damage, post-intubation croup, and, in severe cases, subglottic stenosis. Although the pediatric larynx is conical, the narrowest part, the rima glottidis, is cylindrical in the anteroposterior dimension, regardless of development, and the cricoid ring is slightly elliptical. A cuffed ETT reduces the number of endotracheal intubation attempts, and if cuff pressure can be maintained within a safe range, the risk of airway damage may not be greater than that of an ETT without cuff. The age-based formula suggested by Cole (age/4 + 4) has long been used to select the appropriate ETT size in children. Because age-based formulas in children are not always accurate, various alternative methods for estimating the ETT size have been examined and suggested. Chest radiography, ultrasound, and a three-dimensional airway model can be used to determine the appropriate ETT size; however, there are several limitations.
INTRODUCTION
Selection and insertion of an endotracheal tube (ETT) of appropriate size for airway management during general anesthesia in pediatric patients is an essential, albeit challenging task. The outer diameter of the ETT is very important to control leakage and protect from aspiration, while the lumen of the ETT is important for ventilation pressure. The use of the inner diameter to estimate the optimal ETT size has been established clinically. Although traditional age- or height-based formulas have been used to determine the ETT size [1], the initial size selected may not be appropriate, and replacement may be required. If a very small ETT is inserted, there is a risk of inadequate ventilation and decreased reliability of end tidal carbon dioxide monitoring. There is also a risk of air leakage, anesthetic gas leakage, aspiration, and high airway resistance [2–5]. Similarly, a very large ETT may cause serious complications including airway damage, such as ulceration, local ischemia, scar formation, post-intubation croup, and, in severe cases, subglottic stenosis [6,7]. Therefore, this review article aims to discuss the advantages and disadvantages of conventional and novel methods that are used to select an appropriate ETT size in pediatric patients.
PEDIATRIC AIRWAY ANATOMY
The larynx is mainly funnel-shaped during infancy, with the narrowest part being the nondistensible cricoid cartilage. According to Eckenhoff [8], the larynx is located between the pharynx and trachea, and extends from the base of the tongue to the cricoid cartilage. It consists of the thyroid cartilage, cricoid cartilage, paired arytenoids, epiglottis, and corniculate and cuneiform cartilages located around the epiglottis. These pieces of cartilage are connected with ligaments and affect laryngeal muscle movement. Among them, the cricoid cartilage is located below the largest thyroid cartilage at the C4 level at birth, before settling at the C5 level by 6 years of age [9]. Owing to the small size of the cricoid cartilage in children, mucosal edema may cause severe airway obstruction, and prolonged endotracheal intubation increases the risk of subglottic stenosis [10]. The epiglottis is wide and runs parallel to the trachea in adults, but is narrower, softer, and relatively horizontal in children [11].
Recently, in vivo investigations and review articles have examined whether the larynx is funnel-shaped in children. Holzki et al. [12] reported that when the aryepiglottic, vestibular, and vocal folds, which constitute the structure of the laryngeal upper airway, are maximally abducted, they have a larger opening in the glottis than the outlet of the cricoid ring. Moreover, although the pediatric larynx is conical, the narrowest part, the rima glottidis, is cylindrical in the anteroposterior dimension regardless of development, and the cricoid ring is slightly elliptical [13,14]. However, based on the active contraction of the laryngeal muscles, Litman et al. [15] suggested that the narrowest part of the larynx is the transverse dimension at the level of the vocal cords, while the cricoid ring is functionally the narrowest part.
UNCUFFED VS. CUFFED ETTS
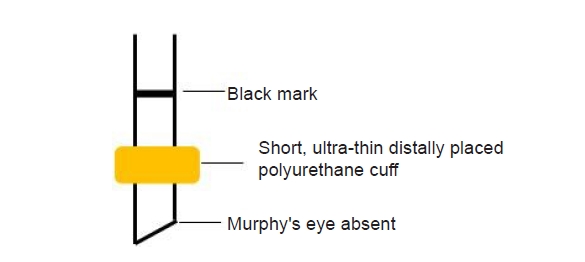
Uncuffed ETTs and cuffed ETTs have advantages and disadvantages (Table 1) [13]. Traditionally, uncuffed ETTs have been used in children below 8 years old, because the cricoid cartilage is the narrowest part in this age range [16]. Litman et al. [15] and Dalal et al. [17] found that, as the cricoid cartilage is nearly ellipse-shaped, circular uncuffed ETTs may cause leakage in the empty anterior and posterior spaces, thus possibly putting excessive pressure to the sides [14]. Therefore, cuffed ETTs can reduce the number of endotracheal intubation attempts, and if cuff pressure can be maintained within a safe range, the risk of airway damage may not exceed that of an uncuffed ETT [18–20]. In addition, the use of cuffed ETTs reduces the risk of leakage during positive pressure ventilation, ensuring patient safety through adequate ventilation and lack of aspiration, and reduces contamination of the surrounding air by inhaled anesthetics (Fig. 1) [21–23]. Therefore, the use of cuffed ETTs in children is increasing. However, excessive pressure in the cuff can inhibit perfusion to the tracheal mucosa and cause postoperative sore throat, subglottic edema, or stenosis [24–26]. Pediatric ETT cuffs must be very thin to meet the high-volume low-pressure standards, and the cuff must be short enough, so that its upper boundary is stably positioned in the subglottic region below the cricoid cartilage (Fig. 2) [14]. In addition, although the tip of the ETT should have a sufficient safety margin over the tracheal bifurcation, selection of an appropriate ETT is challenging, because the outer diameter, size, and location of the cuff vary between manufacturers. The cuff may not meet the criteria for high volume and low pressure, and the depth may not be marked [27].

Endotracheal tube (ETT) with cross-sectional circular shape in an elliptical-shaped airway (A) and in a circular airway (B). When placed in an elliptical airway, there may be lateral pressure on the mucosa of the trachea, and possible leakage during continuous positive airway pressure test above and below the ETT in the anterior–posterior diameter. When a cuff (violet) is inflated in the elliptical airway, the airway can be sealed with equal pressure on all aspects of the tracheal wall (C).
Microcuff ETTs are a new type of ETTs, which are of polyurethane. The cuff is located more distally on the ETT shaft, and there is no Murphy’s eye (Fig. 3). Therefore, the cuff may be positioned below the non-distensible cricoid ring, where a 20 cmH2O inflation pressure maintains a cross-sectional area of approximately 150% of the maximal internal tracheal cross-sectional area. In the deflated state, the outer diameter of the microcuff ETT is minimally increased; therefore, it can be safely used in children [28,29]. However, this type of cuff has not been introduced in Korea yet. With the development of newer polyvinyl chloride high-volume low-pressure cuffed ETTs in the late 1990s and ultrathin polyurethane microcuff ETTs in the early 2000s, the use of cuffed ETTs has increased in infants and children weighing ≥ 3 kg. While most studies on cuffed ETTs in infants only included infants ≥ 3 kg [20,21,30–34], Thomas et al. [33] reported that microcuff ETTs can be safely used in neonates weighing < 3 kg.
HOW TO SELECT AN APPROPRIATE ETT SIZE
Conventional methods
The age-based formula suggested by Cole has long been used to select the appropriate ETT size in children [35]. When an uncuffed ETT is used based on the Cole formula, it has a larger internal diameter (ID); therefore, airway resistance, and edema formation caused due to mucosal damage by cuff pressure can be reduced [1]. In children > 2 years old, the Cole formula [ID (mm) =(age/4) + 4.0] can be used to determine the appropriate uncuffed ETT size.
However, a recent study suggested that the pediatric airway shape is elliptical, rather than round [28], leading to a transition from uncuffed to cuffed ETT use in children. In addition, although the ID is standard for each manufacturer, selection of an appropriate cuffed ETT size is difficult, because the outer diameter varies [29,36]. Accordingly, Khine et al. [31] suggested a new formula [ID (mm) =(age/4) + 3.0] for the increased outer diameter of a cuffed ETT because of the inflatable cuff. However, Duracher et al. [37] recommended a novel formula [ID (mm) =(age/4) + 3.5] to select the appropriate cuffed ETT size, because Khine’s formula was found to underestimate the ETT size by 0.5 mm. Recently, Manimalethu et al. [38] compared these three formulas and found that Duracher’s formula best determined the appropriate size when a cuffed ETT was used. However, application of this formula in infants weighing less than 3 kg and children under 2 years old is difficult. Khine et al. [31], Motoyama [16] and Salgo et al. [39] suggested the use of cuffed ETT sizes according to age (Table 2).
Newly induced method
Because age-based formulas in children are not always accurate, ETTs with an inner diameter with a 0.5 mm deviation from the calculated size for endotracheal intubation should be prepared [1,40]. In cases of an inappropriate ETT size, unnecessary endotracheal intubation may be attempted several times. Various methods for estimating ETT size, other than the age-based formula, have been examined and suggested.
1. Chest radiography
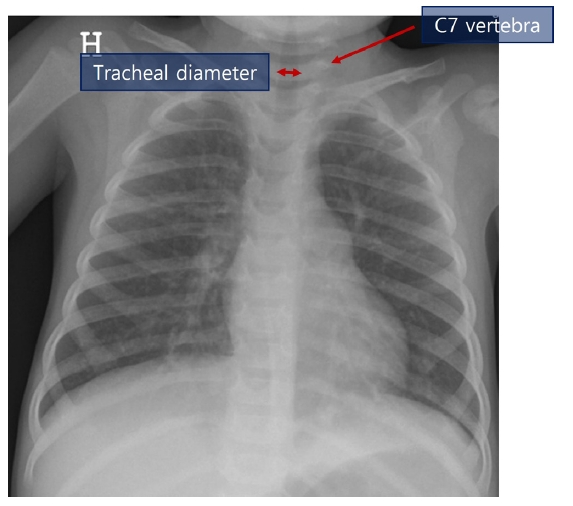
Park et al. [41] conducted a study using uncuffed ETTs in 537 pediatric patients between 3 and 6 years old and recommended a radiograph-based formula [ID (mm) = 3 + 0.3 × (tracheal diameter at seventh cervical vertebra)] through linear regression modeling. The authors measured the tracheal diameter at the seventh cervical vertebra based on chest radiography (Fig. 4) and substituted it into the radiograph-based formula. The success rate was higher than that of the standard age-based formula [ID (mm) = (age/4) + 4.0].
2. Ultrasound
Ultrasound is a reliable, noninvasive, point-of-care test that can be used to evaluate the airway diameter. Recently, several studies have investigated the use of ultrasound in ETT size selection in children (Table 3) [42–46]. Altun et al. [47] reported that the subglottic diameter of the airway, measured with ultrasound (Fig. 5) to determine the appropriate size of cuffed ETT for children, can be a reliable parameter. In addition, the use of ultrasound for airway evaluation in children can provide information about subglottic airway narrowing and vocal cord movement and help to predict a difficult airway [48].
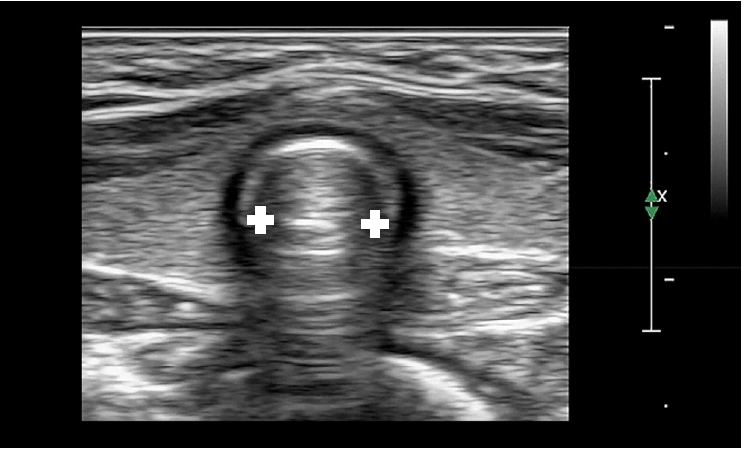
Ultrasonographic measurement of the transverse subglottic diameter of the cricoid cartilage. The cricoid cartilage appears as a round hypoechoic structure with hyperechoic edges, composed of perichondrium. The transverse subglottic diameter is determined by measuring the dimension of the air-mucosa interface (between the two white crosses) within the bilateral inner margin of the cricoid cartilage.
However, because the ultrasound-measured airway diameter represents the outer diameter of the ETT, it must be converted to the corresponding inner diameter. Therefore, the ETT size may vary owing to the different outer diameters used between manufacturers. Reportedly, the subglottic anteroposterior diameter in children is slightly larger than the transverse diameter [15,28], the assessment of anteroposterior diameter is limited due to air shadows, and the measurement of transverse diameter at this level may underestimate the actual tracheal diameter [42].
3. Three-dimensional (3D) airway model
Park et al. [49] found that prediction of the correct ETT size using a 3D-printed airway model demonstrated better results than the age-based formula in pediatric patients with congenital heart disease. This shows that selection of ETT size using a 3D-printed airway model may be feasible to reduce the number of reintubation attempts and complications. However, image quality can be affected by patient respiration during computed tomography (CT) scans, and it is difficult to apply 3D conversion software programs to airway images, compared to solid organs, because of the air layer. In addition, image conversion is required through the use of several programs and CT (Fig. 6); however, it cannot be used to quickly determine the size of the ETT because of the time required implementation of 3D image. It is also limited in its ability to identify a difference from the proper size of the ETT, because it is less compliant after printing and fixing at room temperature.

(A, left) An FDM type 3D printer CreatBot (CreatBot F430, Henan Suwei Electronic Technology Co., Ltd., China) was used to print 3D airway models from the sub-glottis to the upper carina. (A, middle) 3D-printed airway model. (A, right). Cuffed ETTs is inserted into a 3D-printed airway model. (B) DICOM files of pediatric patient’s CT images were converted to STL files using the 3D conversion software open source program InVersalius (InVersalius 3.0, Renato Archer Information Technology Center, Brazil). (C) STL files were converted to G-Code 3D Printer File [49]. Adapted from the article of Park et al. (Korean J Anesthesiol 2021;74:333-41) [49]. FDM: fused deposition modeling, DICOM: Digital Imaging and Communications in Medicine, CT: computed tomography, ETT: endotracheal tube, STL: Standard Triangle Language, 3D: three-dimensional.
CONCLUSION
The age-based formula presented by Cole [ID (mm) = (age/4) + 4.0] has long been used to select the appropriate ETT size in children. This formula is suitable for uncuffed ETTs; however, recent changes in the understanding of pediatric airway anatomy, redefined the use of cuffed ETTs. Cuffed ETTs can reduce the number of endotracheal intubation attempts, and if cuff pressure can be maintained within a safe range, the risk of airway damage may not increase, compared to an endotracheal tube without a cuff. When estimating cuffed ETT size using an age-based formula, Duracher’s formula [ID (mm) = (age/4) + 3.5] is more accurate. However, a tube 0.5 mm larger or smaller than the calculated size should always be possible to use. Because age-based formulas in children are not always accurate, various methods for estimating ETT size, such as chest radiography, ultrasound, and three-dimensional airway models, have been investigated. However, these methods have several limitations, and further studies are warranted to ensure proper use in clinical situations.
Notes
FUNDING
This work was supported by a 2022 research grant from Pusan National University Yangsan Hospital.
CONFLICTS OF INTEREST
Hee Young Kim has been an editor of the Anesthesia and Pain Medicine since 2022: However, she was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
DATA AVAILABILITY STATEMENT
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
AUTHOR CONTRIBUTIONS
Conceptualization: Seyeon Park, Hee Young Kim. Data curation: Seyeon Park, Hee Young Kim. Formal analysis: Hee Young Kim. Writing - original draft: Seyeon Park, Sang-Wook Shin, Hye-Jin Kim, Ji-Uk Yoon, Gyeong-Jo Byeon, Eun-Jung Kim, Hee Young Kim. Writing - review & editing: Seyeon Park, Sang-Wook Shin, Hye-Jin Kim, Ji-Uk Yoon, Gyeong-Jo Byeon, Eun-Jung Kim. Investigation: Hee Young Kim. Supervision: Seyeon Park, Hee Young Kim. Validation: Seyeon Park, Hee Young Kim.