|
 |
- Search
| Anesth Pain Med > Volume 17(3); 2022 > Article |
|
Abstract
Background
This study aimed to evaluate the postoperative analgesic effect of magnesium sulfate during abdominal surgery.
Methods
This randomized double-blinded study involved 84 patients candidates for abdominal surgery into two same groups. In the magnesium group, at first 25 mg/kg/1 h magnesium sulfate; and then, 100 mg/kg/24 h was infused in the intensive care unit. The pain intensity (the primary outcome), was assessed using the numeric rating scale (NRS) every 3 h. If the NRS was > 3, morphine (as a secondary outcome) was used and evaluated. The results were analyzed using SPSS ver. 19 software, and statistical significance was set at P < 0.05
Results
Demographic parameters were similar between the groups. The pain intensity were similar at first and then at the third hour in both groups (P = 0.393 and P = 0.172, respectively), but thereafter between 6 and 24 h, the pain severity was significantly lower in the magnesium group (4.4 ± 1.3 in the control and 3.34 ± 1 in the magnesium group at 6th hour and P = 0.001). In addition, morphine intake in the first 24 h in the two groups had a significant difference, with 13.2 ± 5.7 mg in control group and 8 ± 3.5 mg in magnesium group (P = 0.001).
Postoperative pain can cause many complications such as restlessness, increased sympathetic activity, high blood pressure, and tachycardia. Opioids are effective analgesics; however, they produce unwanted side effects, such as respiratory depression, nausea, and vomiting. Nonopioid drugs can decrease opioid usage and dependency [1,2]. Postoperative pain management improves the patient’s recovery time and activity, and reduces the risk of thromboembolism. Furthermore, pain control leads to faster discharge in outpatient surgeries and is one of the most critical parameters in the enhanced recovery after surgery protocol [3].
According to numerous reports, magnesium sulfate has analgesic effects and lowers opioid consumption [4-9]. Magnesium inhibits calcium channels and N-methyl-D-aspartate (NMDA) receptors for pain relief; however, the mechanism is unknown [10]. Magnesium sulfate is used for local and regional anesthesia, intravenous injections, and oral consumption [11-13]. However, magnesium can prolong muscle relaxation, sedation, slow recovery time, dysrhythmia, and negative inotropic effects [5,14].
In some studies, the side effects of magnesium were higher than the pain-relieving effects; therefore, the analgesic effects require further evaluation [10,13]. In this study, we hypothesized that magnesium sulfate would reduce postsurgical pain and opioid consumption; therefore, we evaluated the effect of magnesium sulfate on pain relief and opioid usage in abdominal surgery.
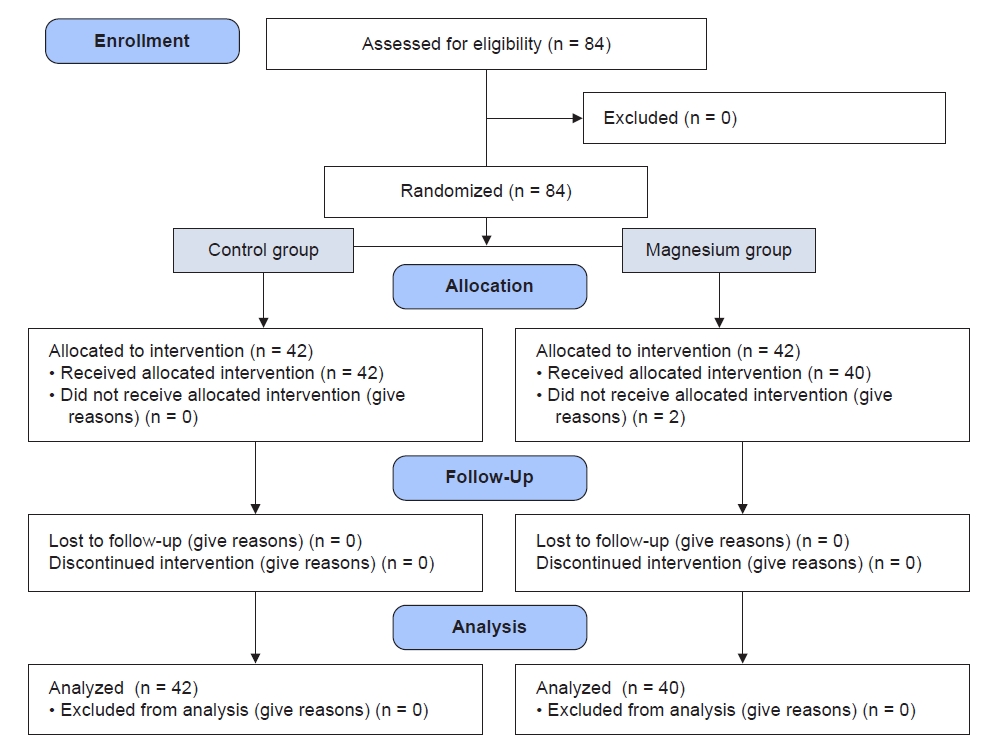
This study is a randomized double-blinded clinical trial conducted in the surgical intensive care unit (ICU) of the university’s educational center. The local ethical committee approved this study (no. IR.MUMS.MEDICAL.REC.980757 on 5/11/2019), and it was registered in the Iranian registration center (no. IRCT20210214050359N1 on 24/8/2021). In this study, 84 patients who were candidates for major cancer abdominal surgery with open laparotomy and American Society of Anesthesiologists physical status I-II were evaluated. After acquiring the patients’ consent forms, they were randomly assigned to two groups (magnesium sulfate and control; allocation ratio 1:1) using an online random number table. In this study, 84 patients were recruited, but two patients in the magnesium group refused to follow up (n = 2). The remaining 82 patients fulfilled all the criteria and participated in the study. Each patient was allocated to a group using a sealed envelope. Patients and nurses who assessed the patients were blinded to the study groups.
Patients with magnesium sulfate allergy, opioid dependency, addiction, neuromuscular disease, low ejection fraction (< 40%), liver and kidney failure, and dissatisfaction with participating in the study were excluded.
All the patients were monitored for blood pressure, electrocardiography, oxygen saturation, and capnography. In both groups, the patients were induced with fentanyl (2 µg/kg), 1 mg of midazolam, propofol (2-2.5 mg/kg), and atracurium (0.5 mg/kg). Anesthesia was maintained by propofol infusion (100-150 µg/kg/min) and fentanyl bolus (1 mg/kg) every half hour. After the surgery, neostigmine (0.04 mg/kg) and atropine (0.02 mg/kg) were administered, endotracheal tube was removed, and transfer to the recovery unit was done. Muscle relaxation status was carefully evaluated using a 5-s head lift and grip strength maneuver before injecting magnesium sulfate when they were awake. In the magnesium group, 25 mg/kg of magnesium sulfate was injected for 1 h. After ICU transfer, magnesium sulfate was infused with 100 mg/kg/d saline solution. In the control group, 15 ml/L sterile distilled water was added to isotonic saline as placebo, and infused intravenously. The primary outcome was pain intensity, which was evaluated using the NRS (0-10), and the secondary outcome was opioid consumption, which was observed every 3 h in 24 h period at the ICU. If the NRS was > 3, morphine (0.02 mg/kg) was used to reduce pain. No other analgesics were prescribed during the study period. In addition, all patients were monitored for magnesium side effects, such as deep tendon reflex and respiratory rate, every 3 h. Magnesium injection was stopped if any side effects such as bradypnea and muscle relaxation were observed.
Using the G*Power V3.1 software and according to Kashefi et al. [15], with an average NRS of 3.3, a reduction of 30%, an alpha error 0.01 and 90% power, a sample size of 34 persons in each group was obtained. Considering a 20% decline, 42 individuals were chosen from each group (a total of 84 participants). SPSS ver. 19 software (IBM Co., USA) was used for data analysis, and a P value < 0.05 was significant. Chi-square and Fisher exact tests were used to compare non-parametric data, and t-tests, Mann-Whitney tests, and repeated measures analysis of variance (ANOVA) were used to compare parametric data, such as NRS and morphine dosage.
In total, 84 patients (42 in each group) participated in this study from June 2021 to May 2021 in the surgical ICU of the university hospital. Two patients in the magnesium group were excluded from the study due to follow-up refusal (Fig. 1). Demographic data, such as age, sex, American Society of Anesthesiologists physical status, BMI, and duration of surgery, were similar in both groups (Table 1).
The pain levels were evaluated using the NRS (0-10) every 3 h. After 6 h, there was a significant difference in pain intensity between the two groups (Table 2). At the beginning of the study, the NRS levels were 4 ± 0.2 in the control group and 3.8 ± 0.2 in the magnesium group (P = 0.393). After 3 h, the NRS levels were 3.98 ± 0.2 in the control group and 3.45 ± 0.2 in the magnesium group (P = 0.172). In subsequent evaluations, the pain intensity in the magnesium group was significantly lower than that in the control group (Fig. 2).
Additionally, the morphine intake after 1 h and 3 h post-operation did not have a significant difference (P= 0.950, 0.451 respectively). However, morphine intake in the magnesium group was lower than that in the control group after 6 h. Subsequently, morphine dosage was lower in the magnesium group in the first 24 h (13.2 ± 5.7 mg in the control group and 8 ± 3.5 mg in the magnesium group (P = 0.001). No considerable changes were observed in the hemodynamic condition, respiratory rate (P = 0.759), muscle relaxation, and deep tendon reflex (P = 0.322).
This study demonstrated that magnesium sulfate reduced pain levels after 3 h in a major abdominal surgery, and there were no unwanted side effects. In the first 3 h, no significant difference was observed between the two groups, which may be due to the persistence of the intraoperative analgesic effect and peak delay effect of magnesium sulfate. In addition, it reduced the need for opioids, 24 h after surgery. In our previous study, hypomagnesemia prevalence was 46.7% in hospitalized patients in the ICU after the initial 24 h of elective surgery [16]. A single dose of magnesium to correct hypomagnesemia may decrease pain intensity.
Magnesium is a physiological inhibitor of calcium channels; however, its mechanism of pain relief remains unknown. It may also act as an analgesic by adjusting the flow of calcium into the cell and may also serve as an antagonist to the NMDA receptor. On the other hand, intraoperative magnesium injection can lead to dangerous side effects, such as prolonged muscle relaxant effects, prolonged recovery, and sometimes severe heart disturbances. The effects of magnesium on the bispectral index (BIS) and sedation were also studied, and it was shown that magnesium could significantly reduce BIS [12,14].
Magnesium sulfate has been used for analgesia in various surgeries, such as hysterectomy, abdominal surgery, orthopedics, and the thorax, which reduces pain levels and opioid usage [17-21]. Magnesium is used as a loading dose or infusion for regional anesthesia, intra-articular injection, and oral administration after surgery [11-13]. Intra-articular injection of magnesium after arthroscopy helped patients to move faster and reduce their pain [22]. Oral consumption of magnesium results in reduced pain and trismus of the jaw muscles after tooth removal [11]. In a meta-analysis study, no significant difference was observed in the various methods of prescribing magnesium and its effectiveness in pain relief. In addition, no difference was observed between total magnesium consumption and reduced morphine intake during the initial 24 h [23].
In a study conducted by Mehraeen et al., pre-anesthesia injection of 25 and 50 mg/kg of magnesium sulfate did not reduce pain levels during inguinal hernia surgery [24]. Subsequently, in another study conducted by Wilder-Smith et al. [25], magnesium injection before hysterectomy did not affect postoperative pain levels. In a study on knee arthroscopy, the bolus and infusion of magnesium sulfate were not adequate for pain relief [26]. In a study on children, magnesium injection did not affect pain levels and sedation [27]. The difference between these findings and those of our study may be due to the type of surgery, time, method, and magnesium dosage.
In this study, we evaluated the effects of magnesium on hemodynamic and muscle relaxation. No significant side effects, such as muscle relaxation or cardiovascular complications, were observed during the initial 24 h of the surgery. No significant differences were observed between studies on magnesium side effects such as muscle relaxation and hypotension [23]. However, in some studies, bradycardia was more common in the magnesium group but without hemodynamic instability [28,29]. In a research conducted on 10,000 pregnant women with preeclampsia, no side effects from high doses of magnesium (28 g) were observed in the first 24 h [30].
One of the limitations of this study was the small sample size. Also, the pain control methods were applied only in painful and complicated abdominal surgeries. Consequently, the study included ICU patients who required more sedatives.
In conclusion, prescribing magnesium in the form of bolus and infusion in the ICU can reduce pain levels and morphine dosage, during the first 24 h after abdominal surgery, which does not result to any significant complications. However, owing to these limitations, further evaluation is necessary to determine the effectiveness of magnesium sulfate in pain control.
Notes
DATA AVAILABILITY STATEMENT
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
AUTHOR CONTRIBUTIONS
Conceptualization: Arash Peivandi Yazdi. Data curation: Arash Peivandi Yazdi. Formal analysis: Mehrdad Esmaeeli. Funding acquisition: Mehrdad Esmaeeli. Methodology: Arash Peivandi Yazdi, Mehrdad Esmaeeli. Project administration: Mehrdad Esmaeeli. Visualization: Mehrdad Esmaeeli. Writing - original draft: Mehryar Taghavi Gilani. Writing - review & editing: Mehryar Taghavi Gilani. Investigation: Mehrdad Esmaeeli. Resources: Arash Peivandi Yazdi. Software: Mehrdad Esmaeeli. Supervision: Mehrdad Esmaeeli. Validation: Arash Peivandi Yazdi.
Table 1.
Demographic Parameters and Operation Time
Table 2.
Pain Intensity at 24 h on Numeric Rating Scale (NRS)
REFERENCES
1. Wu CL, Fleisher LA. Outcomes research in regional anesthesia and analgesia. Anesth Analg 2000; 91: 1232-42.


2. Camu F, Van Aken H, Bovill JG. Postoperative analgesic effects of three demand-dose sizes of fentanyl administered by patient-controlled analgesia. Anesth Analg 1998; 87: 890-5.


3. Kehlet H, Wilmore DW. Multimodal strategies to improve surgical outcome. Am J Surg 2002; 183: 630-41.


4. Buvanendran A, McCarthy RJ, Kroin JS, Leong W, Perry P, Tuman KJ. Intrathecal magnesium prolongs fentanyl analgesia: a prospective, randomized, controlled trial. Anesth Analg 2002; 95: 661-6.


5. Fuchs-Buder T, Wilder-Smith OH, Borgeat A, Tassonyi E. Interaction of magnesium sulphate with vecuronium-induced neuromuscular block. Br J Anaesth 1995; 74: 405-9.

6. Bhatia A, Kashyap L, Pawar DK, Trikha A. Effect of intraoperative magnesium infusion on perioperative analgesia in open cholecystectomy. J Clin Anesth 2004; 16: 262-5.


7. Steinlechner B, Dworschak M, Birkenberg B, Grubhofer G, Weigl M, Schiferer A, et al. Magnesium moderately decreases remifentanil dosage required for pain management after cardiac surgery. Br J Anaesth 2006; 96: 444-9.


8. Ryu JH, Kang MH, Park KS, Do SH. Effects of magnesium sulphate on intraoperative anaesthetic requirements and postoperative analgesia in gynaecology patients receiving total intravenous anaesthesia. Br J Anaesth 2008; 100: 397-403.


9. De Oliveira GS Jr, Castro-Alves LJ, Khan JH, McCarthy RJ. Perioperative systemic magnesium to minimize postoperative pain: a meta-analysis of randomized controlled trials. Anesthesiology 2013; 119: 178-90.

11. Albrecht E, Kirkham KR, Liu SS, Brull R. The analgesic efficacy and safety of neuraxial magnesium sulphate: a quantitative review. Anaesthesia 2013; 68: 190-202.


12. Jerkovic D, Tadin A, Gavic L, Vladislavic NZ, Grgic N, Macan D. Effect of orally administered magnesium on postoperative pain level and trismus after surgical removal of the lower third molars: a randomized, double-blind, placebo-controlled trial. Clin Oral Investig 2020; 24: 4649-59.



13. Sane S, Mahdkhah A, Golabi P, Hesami SA, Kazemi Haki B. Comparison the effect of bupivacaine plus magnesium sulfate with ropivacaine plus magnesium sulfate infiltration on postoperative pain in patients undergoing lumbar laminectomy with general anesthesia. Br J Neurosurg. 2020. doi: 10.1080/02688697.2020.1861430. [Epub ahead of print].
14. Gilani MT, Zirak N, Razavi M. Accidental intrathecal injection of magnesium sulfate for cesarean section. Saudi J Anaesth 2014; 8: 562-4.



15. Kashefi P, Montazeri K, Honarmand A, Moradi A, Masoomi SG. Adding magnesium to lidocaine for intravenous regional anesthesia. J Res Med Sci 2008; 13: 108-14.
16. Peyvandi Yazdi A, Hashemi E, Salehi M, Masoumzadeh M, Razavi M. Evaluation of the prevalence of hypomagnesemia in the first 24th hour after selective operations in intensive care unit patients. OFOGH-E-DANESH 2014; 20: 29-33.
17. Moon S, Lim S, Yun J, Lee W, Kim M, Cho K, et al. Additional effect of magnesium sulfate and vitamin C in laparoscopic gynecologic surgery for postoperative pain management: a double-blind randomized controlled trial. Anesth Pain Med (Seoul) 2020; 15: 88-95.



18. Kaya S, Kararmaz A, Gedik R, Turhanoğlu S. Magnesium sulfate reduces postoperative morphine requirement after remifentanil-based anesthesia. Med Sci Monit 2009; 15: PI5-9.

19. Ozcan PE, Tugrul S, Senturk NM, Uludag E, Cakar N, Telci L, et al. Role of magnesium sulfate in postoperative pain management for patients undergoing thoracotomy. J Cardiothorac Vasc Anesth 2007; 21: 827-31.


20. Dabbagh A, Elyasi H, Razavi SS, Fathi M, Rajaei S. Intravenous magnesium sulfate for post-operative pain in patients undergoing lower limb orthopedic surgery. Acta Anaesthesiol Scand 2009; 53: 1088-91.


21. Shin HJ, Kim EY, Na HS, Kim TK, Kim MH, Do SH. Magnesium sulphate attenuates acute postoperative pain and increased pain intensity after surgical injury in staged bilateral total knee arthroplasty: a randomized, double-blinded, placebo-controlled trial. Br J Anaesth 2016; 117: 497-503.


22. Varas V, Bertinelli P, Carrasco P, Souper N, Álvarez P, Danilla S, et al. Intraoperative ketamine and magnesium therapy to control postoperative pain after abdominoplasty and/or liposuction: a clinical randomized trial. J Pain Res 2020; 13: 2937-46.


23. Shi L, Zhu H, Ma J, Shi LL, Gao F, Sun W. Intra-articular magnesium to alleviate postoperative pain after arthroscopic knee surgery: a meta-analysis of randomized controlled trials. J Orthop Surg Res 2021; 16: 111.




24. Albrecht E, Kirkham KR, Liu SS, Brull R. Peri-operative intravenous administration of magnesium sulphate and postoperative pain: a meta-analysis. Anaesthesia 2013; 68: 79-90.


25. Mehraein A, Azad MA, Sadeghi M. The analgesic effect of magnesium sulfate in postoperative pain of inguinal hernia repair. Tehran Univ Med J 2007; 65: 55-8.
26. Wilder-Smith OH, Arendt-Nielsen L, Gäumann D, Tassonyi E, Rifat KR. Sensory changes and pain after abdominal hysterectomy: a comparison of anesthetic supplementation with fentanyl versus magnesium or ketamine. Anesth Analg 1998; 86: 95-101.

27. Salman N, Olgunkeleş B, Bektaş U, Güner D, Bektas M, Ay Ş, et al. [Effects of intraarticular tramadol, magnesium and ketamine on postoperative pain in arthroscopic meniscectomy]. Braz J Anesthesiol 2019 69: 35-41. Portuguese.


28. Lee JH, Choi S, Lee M, Jang YE, Kim EH, Kim JT, et al. Effect of magnesium supplementation on emergence delirium and postoperative pain in children undergoing strabismus surgery: a prospective randomised controlled study. BMC Anesthesiol 2020; 20: 289.




29. Ferasatkish R, Dabbagh A, Alavi M, Mollasadeghi G, Hydarpur E, Moghadam AA, et al. Effect of magnesium sulfate on extubation time and acute pain in coronary artery bypass surgery. Acta Anaesthesiol Scand 2008; 52: 1348-52.


-
METRICS

-
- 2 Crossref
- 4,223 View
- 152 Download
- Related articles in Anesth Pain Med
- ARTICLE & TOPICS
-
- Topics
-
- Neuroscience in anesthesiology and critical care
- Anesthetic Pharmacology
- Obstetric Anesthesia
- Pediatric Anesthesia
- Cardiothoracic and Vascular Anesthesia
- Transplantation Anesthesia
- Spinal Pain
- Regional Anesthesia
- Neuromuscular Physiology and Pharmacology
- Airway Management
- Geriatric anesthesia and Pain
- Others