 |
 |
- Search
| Anesth Pain Med > Volume 17(1); 2022 > Article |
|
Abstract
Background
Excessive citrate load during therapeutic plasma exchange (TPE) can cause metabolic alkalosis with compensatory hypercarbia and electrolyte disturbances. If TPE is required immediately before ABO-incompatible (ABOi) liver transplant (LT) surgery, metabolic derangement and severe electrolyte disturbance could worsen during LT anesthesia.
Case
We report two ABOi LT cases who received TPE on the day of surgery because isoagglutinin titers did not be dropped below 1:8. One case had a surprisingly high metabolic alkalosis with a pH of 7.73 immediately after tracheal intubation because of hyperventilation during mask bagging. The other experienced sudden ventricular tachycardia and blood pressure drop after surgical incision accompanied with severe hypokalemia of 1.8 mmol/L despite supplementation with potassium.
Living-donor ABO-incompatible (ABOi) liver transplantation (LT) is now recognized as the only way to overcome blood type barriers and successfully expand donor pools [1]. However, in case of ABOi LT, hyperacute rejection could be occurred because of immune response by anti-A or anti-B antibodies [2]. Therapeutic plasma exchange (TPE) decreases the naturally occurring anti-A or anti-B isoagglutinin titer. However, TPE, on the morning of LT, is occasionally inevitable in TPE-resistant patients who struggle to lower anti-A or anti-B isoagglutinin titers [3]. The isoagglutinin titers should be dropped below 1:8 to avoid hyperacute rejection [4,5].
However, an excessive citrate load during TPE can cause hypocalcemia and hypomagnesemia and acid-base imbalances [6]. Therefore, metabolic alkalosis with compensatory hypercarbia and electrolyte disturbances occurred before LT [7,8]. If TPE was performed the day before surgery, patients may have time to correct acid-base imbalance and electrolyte disturbances by respiratory compensation or supplement of electrolytes. On the contrary, TPE was performed even on the day of surgery, this imbalance will be further exacerbated, which can cause severe arrhythmias and fatal outcomes.
We report on two ABOi LT cases including severe metabolic alkalosis and ventricular tachycardia who received TPE on the day of surgery. One case had a surprisingly high metabolic alkalosis of pH 7.73 immediately after tracheal intubation and the other had sudden ventricular tachycardia after surgical incision accompanied with severe hypokalemia of 1.8 mmol/L.
These case reports were approved by the Institutional Review Board (no. 2021-0691).
A 58-year-old male with alcoholic cirrhosis (Model for End-Stage Liver Disease [MELD] score: 17) received ABOi LT with TPE on the operative morning. The initial isoagglutinin titer of the patient was 1:2,048, and even after TPE was performed eight times up to the day before surgery, the titer was 1:32. The 9th TPE was performed on the morning of surgery, with 29 units (replaced volume 4,759 ml) of fresh frozen plasma (FFP) replaced and an acid-citrate-dextrose solution used as an anticoagulant. LT was then performed, with the final titer of 1:8.
Room air arterial blood gas analysis (ABGA) (Table 1) was performed after entering the operating room. The arterial partial pressure of carbon dioxide (PaCO2) and bicarbonate (HCO3) were unexpectedly high and were reexamined after encouraging deep breathing. Although we tried to avoid an unintentional increase of minute ventilation during anesthesia induction and manual bagging, which may disrupt compensatory hypercarbia, we found that PaCO2 was decreased from 75 mmHg to 46 mmHg. Consequently, this caused uncompensated severe metabolic alkalosis (pH, 7.53 to 7.73) with sustained HCO3 retention (> 60 mmol/L). Severe hypokalemia (2.1 mEq/L) was also noted.
A 39-year-old male with alcoholic cirrhosis (MELD score: 18) received ABOi LT with an 8th TPE the operative morning. The initial isoagglutinin titer of the patient was 1:4,096. The titer was 1:16, even though TPE was performed seven times until the day before surgery. Therefore, the 8th TPE was performed on the morning of surgery, with 20 units (replaced volume 3,287 ml) of FFP replaced and an acid-citrate-dextrose solution used as an anticoagulant. LT was then performed with the final titer of 1:8.
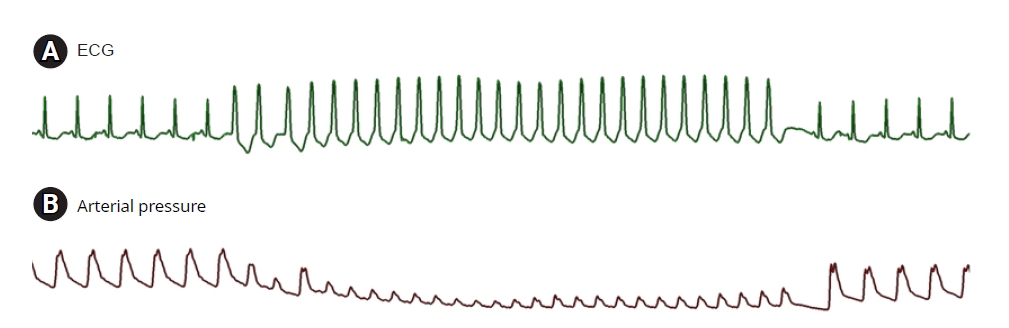
Metabolic alkalosis was revealed on room air ABGA (Table 2) after entering the operating room. Anesthesia was induced, with careful attention to avoid hyperventilation by bag and mask ventilation. After anesthesia induction, metabolic alkalosis and respiratory compensation were shown on ABGA with severe hypokalemia (1.8 mEq/L). Potassium chloride was supplemented, and hypokalemia was slightly resolved. However, ventricular tachycardia with severe blood pressure drop occurred for 10 s about 40 min after the incision, but the patient spontaneously recovered (Fig. 1).
According to the World Health Organization and the Spanish Transplant Organization, Organizaci├│n Nacional de Trasplantes Global Observatory on Donation and Transplantation, more than 30,000 LTs per year are performed worldwide as a potential curative treatment for patients with end-stage liver disease or hepatocellular carcinoma. However, a donor organ shortage in Korea has led to high waiting lists and increased mortality rates. Therefore, to overcome organ shortage, living-donor ABOi LTs have become increasingly popular [9,10].
Historically, ABOi LT has been challenged with various desensitization methods, such as TPE, direct graft infusion therapy, cyclophosphamide, splenectomy, and intraarterial or intravenous immunoglobulin therapies [2,8]. Currently, desensitization protocol of TPE with preemptive rituximab administration has been standardized and has allowed ABOi living donor liver transplantation (LDLT) to become more successful [1-3].
Pre-LT TPEs are typically performed many times until the day before LT to effectively decrease anti-A or anti-B isoagglutinin titers until dropped below 1:8 [4,5]. The titers were followed up daily before liver transplantation to ensure that the titer decreased below 1:8. Through the procedure, the incidence of hyperacute rejection in ABOi LT can be reduced. Usually, TPE was performed using a dual lumen central venous catheter in the internal jugular vein. The infusion rate was set at approximately 40 ml/min and was adjusted according to the patient's tolerance. Albumin, which role a significant part in blood osmotic pressure, was the preferred replacement fluid as long as coagulation lab test was within the normal range [11]. Regional citrate anticoagulation (RCA) was infused with a huge amount of FFP as replacement fluid. The FFP infused approximately 1.5 times the plasma volume [4]. The FFP can compensate for the loss of coagulation factors accompanied by plasma drainage. The RCA used in TPE, which was combined with ionized calcium, which was a necessary cofactor of the coagulation cascade [12]. Accumulation of citrate causes systemic hypocalcemia, which leads to tetany, QT prolongation and life-threatening arrhythmias may occur [13]. Therefore, during TPE, calcium gluconate was continuously infused to prevent hypocalcemia [14]. However, the massive citrate load from the RCA solution and FFPs might shift the acid-base status in liver cirrhosis patients from respiratory alkalosis to metabolic alkalosis [8]. Consequently, with pre-existing electrolyte disturbances, the risk of severe hypokalemia and hypomagnesemia are aggravated in ABOi LDLT recipients [8].
Therefore, anesthesiologists should be alert in patients with ABOi LT because inadvertent hyperventilation might take place during anesthesia induction with manual bagging. Such hyperventilation may lead to severe alkalosis because rapid respiratory alkalosis initiated by an unintentional fall in PaCO2 profoundly aggravates preexisting metabolic alkalosis by TPE [8].
In our first case, we were surprised to find that a pH of 7.7 and severe hypokalemia after tracheal intubation developed, even though we intended to avoid hyperventilation. This implies we should have paid more attention and been more careful during manual bagging. After finding this severe alkalosis, we decreased minute ventilation to compensate severe alkalosis and small amount of potassium chloride was supplemented.
Our second case developed ventricular tachycardia with severe hypokalemia (1.8 mmol/L) after surgical incision. Fortunately, the ventricular tachycardia running time was short and recovered spontaneously without treatment. We therefore could begin to manage the acid-base and electrolyte disturbances without further ventricular arrhythmias.
PotassiumŌĆÖs the arrhythmogenic potential on the heart has been extensively studied [15]. If severe, it is associated with life-threatening ventricular arrhythmias. Various literatures demonstrated a significant positive correlation between hypokalemia and the incidence of malignant ventricular arrhythmias. Therefore, in case of severe hypokalemia, potassium supplementation should be actively carried out within the recommended infusion rate.
The electrocardiographic (ECG) criteria for hypokalemia include the presence of U waves greater than 1 mm and U waves larger than the T wave with associated ST-segment depression [15]. Before developing ventricular tachycardia, our second case of hypokalemia also showed similar hypokalemic ECG changes.
In conclusion, our cases report strongly suggests that anesthesiologists should pay particular attention to PaCO2 changes and hypokalemia in ABOi LT, particularly at the start of anesthesia and mechanical ventilation in patients who just finished TPE the operative day morning. This may disturb the acid-base homeostasis and electrolyte balance, resulting in life-threatening ventricular tachycardia.
Notes
Fig.┬Ā1.
(A) ECG and (B) arterial pressure before and after ventricular tachycardia. Just before ventricular tachycardia, hypokalemic ECG findings, such as ST-segment depression, T-wave flattening, and prominent U waves were seen. ECG: electrocardiography.
Table┬Ā1.
Artery Blood Gas Analysis after Entering the Operating Room
FiO2: fraction of inspired oxygen, EtCO2: end-tidal carbon dioxide, pH: potential of hydrogen, pCO2: partial pressure of carbon dioxide, pO2: partial pressure of oxygen, HCO3: bicarbonate, BE: base excess, Sat: oxygen saturation, Na: sodium, K: potassium, Ca: calcium, Lac: lactate, Hct: hematocrit, intu.: intubation, inci.: incision, ?: uncheckable.
Table┬Ā2.
Artery Blood Gas Analysis after Entering the Operating Room
FiO2: fraction of inspired oxygen, EtCO2: end-tidal carbon dioxide, pH: potential of hydrogen, pCO2: partial pressure of carbon dioxide, pO2: partial pressure of oxygen, HCO3: bicarbonate, BE: base excess, Sat: oxygen saturation, Na: sodium, K: potassium, Ca: calcium, Lac: lactate, Hct: hematocrit, intu.: intubation, min.: minutes, VT: ventricular tachycardia.
REFERENCES
1. Yoon YI, Song GW, Lee SG, Hwang S, Kim KH, Kim SH, et al. Outcome of ABO-incompatible adult living-donor liver transplantation for patients with hepatocellular carcinoma. J Hepatol 2018; 68: 1153-62.


2. Song GW, Lee SG, Hwang S, Kim KH, Ahn CS, Moon DB, et al. Biliary stricture is the only concern in ABO-incompatible adult living donor liver transplantation in the rituximab era. J Hepatol 2014; 61: 575-82.


3. Kozaki K, Egawa H, Kasahara M, Oike F, Yoshizawa A, Fukatsu A, et al. Therapeutic strategy and the role of apheresis therapy for ABO incompatible living donor liver transplantation. Ther Apher Dial 2005; 9: 285-91.


4. Roman PE, DeVore AD, Welsby IJ. Techniques and applications of perioperative therapeutic plasma exchange. Curr Opin Anaesthesiol 2014; 27: 57-64.


5. Shunkwiler SM, Pham HP, Wool G, Ipe TS, Fang DC, Biller E, et al. Therapeutic Apheresis Subsection of the AABB. The management of anticoagulation in patients undergoing therapeutic plasma exchange: a concise review. J Clin Apher 2018; 33: 371-9.


6. Kramer L, Bauer E, Joukhadar C, Strobl W, Gendo A, Madl C, et al. Citrate pharmacokinetics and metabolism in cirrhotic and noncirrhotic critically ill patients. Crit Care Med 2003; 31: 2450-5.


7. Mokrzycki MH, Kaplan AA. Therapeutic plasma exchange: complications and management. Am J Kidney Dis 1994; 23: 817-27.


8. Kwon HM, Jun IG, Lee J, Moon YJ, Jung KW, Jeong HW, et al. Prevalent metabolic derangement and severe thrombocytopenia in ABO-incompatible liver recipients with pre-transplant plasma exchange. Sci Rep 2018; 8: 6679.



9. Kim JM, Kwon CH, Joh JW, Kang ES, Park JB, Lee JH, et al. ABO-incompatible living donor liver transplantation is suitable in patients without ABO-matched donor. J Hepatol 2013; 59: 1215-22.


10. Kwon HM, Hwang GS. Cardiovascular dysfunction and liver transplantation. Korean J Anesthesiol 2018; 71: 85-91.



11. Pandey P, Setya D, Sinha V, Bhatt A, Devra A, Chaudhary A, et al. Therapeutic apheresis in ABO-incompatible kidney and liver transplantation: a single-center experience of 50 patients. Ther Apher Dial 2021; 25: 103-17.


12. Salvadori M, Tsalouchos A. Therapeutic apheresis in kidney transplantation: an updated review. World J Transplant 2019; 9: 103-22.



- ARTICLE & TOPICS
-
- Topics
-
- Neuroscience in anesthesiology and critical care
- Anesthetic Pharmacology
- Obstetric Anesthesia
- Pediatric Anesthesia
- Cardiothoracic and Vascular Anesthesia
- Transplantation Anesthesia
- Spinal Pain
- Regional Anesthesia
- Neuromuscular Physiology and Pharmacology
- Airway Management
- Geriatric anesthesia and Pain
- Others








