Successful management of uncontrolled postpartum hemorrhage due to morbidly adherent placenta with Resuscitative endovascular balloon occlusion of the aorta during emergency cesarean section - A case report -
Article information
Abstract
Background
Morbidly adherent placenta (MAP) may cause life-threatening postpartum hemorrhage (PPH) requiring massive transfusions. Furthermore, it could endanger the lives of both mother and baby. Despite various efforts, such as adjuvant endovascular embolization and hysterectomy, massive PPH due to MAP still occurs and is difficult to overcome.
Case
Herein, we described the case of a 40-year-old woman with placenta previa totalis who experienced massive bleeding during a cesarean section. We used resuscitative endovascular balloon occlusion of the aorta (REBOA) and it improved the condition of the surgical field and the hemodynamic stability of the patient temporarily. The patient was successfully managed without further complications.
Conclusions
REBOA can be used as a rescue procedure for uncontrolled bleeding situations in patients with MAPs. Anesthesiologists should consider and recommend REBOA as another resuscitative therapeutic option in the case of massive PPH.
Morbidly adherent placenta (MAP), including placenta accreta, increta, and percreta, is characterized by the abnormal invasion of placental tissue into the uterine wall [1,2]. MAP occurs in 1 out of every 333–533 deliveries and is a leading cause of postpartum hemorrhage (PPH) and maternal mortality [3]. It is also associated with an increased risk of major hemorrhage and, thus, with poor maternal outcomes [2,4]. With the increase in the incidence of cesarean sections, there has been an increase in the number of MAP cases as well [5,6].
Traditionally, hysterectomy following cesarean section is usually indicated as the treatment option for patients with MAPs. However, to minimize intraoperative hemorrhage and facilitate surgery, some new treatment options have been described, including ligation of the internal iliac artery or uterine artery and placement of a balloon catheter in the iliac artery, uterine artery, or even in the aorta [7,8]. Despite efforts like these adjuvant strategies, massive bleeding due to MAPs still occurs and is difficult to overcome.
Resuscitative endovascular balloon occlusion of the aorta (REBOA) is a minimally invasive procedure that introduces a balloon occlusion catheter into the aorta to achieve endovascular aorta occlusion [9]. It is a life-saving procedure used to control bleeding and maintain blood pressure temporarily in traumatic hemorrhagic shock. Recently, REBOA has been used in postpartum hemorrhage caused by MAP and its efficacy has also been investigated [10]. However, no case of the use of REBOA to control bleeding in PPH has been reported yet in Korea.
Here, we described a case where REBOA was used to treat a life-threatening hemorrhage in a patient with MAP.
CASE REPORT
Written informed consent was obtained from the patient for the presentation and publication of this case report.
A 40-year-old, 56-kg Korean woman (gravida 4, para 2) was scheduled for an emergency cesarean section at 35 weeks and 3 days of gestation for preterm labor. Her medical history included one abortion and two prior cesarean sections for placenta previa.
In the preoperative laboratory tests, her hemoglobin (Hb) was 11.4 g/dl and platelet count was 247,000/μl. Other laboratory findings, including coagulation tests, were within the normal range. On her ultrasound examination, the placenta was suggestive of placenta previa totalis, with possible placenta accreta (Fig. 1).
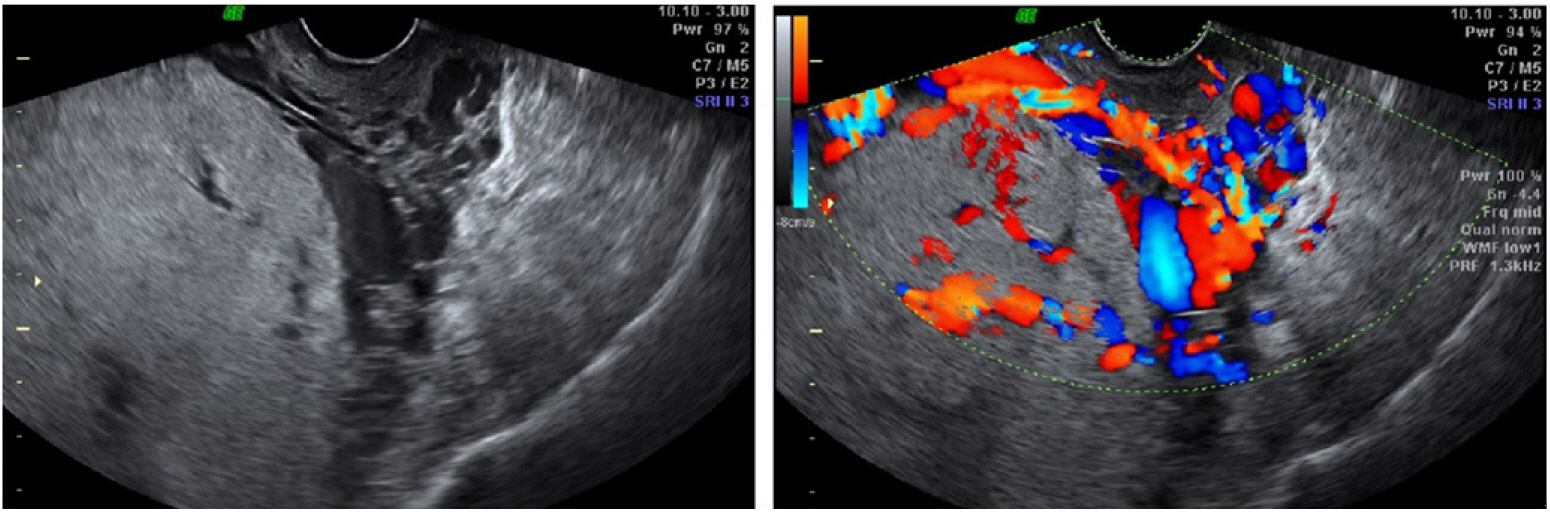
Preoperative ultrasonography findings of the patient’s placenta indicating suspicious placenta previa with accreta.
In the operating room, spinal anesthesia was performed at the L3–4 intervertebral space of the patient. After confirming the spontaneous flow of cerebrospinal fluid, 10 mg of 0.5% heavy bupivacaine and 10 μg of fentanyl were intrathecally injected using a 27-gauge (G) needle. The maximum sensory blockade level of anesthesia was T4. To prepare for arterial blood pressure monitoring, a 22 G angiocatheter was inserted into the right radial artery. Two 18 G intravenous lines were secured in both arms.
Three minutes after starting the operation, a female baby (2,590 g) was delivered with Apgar scores of 7 and 8 at 1 and 5 min, respectively. After the baby was delivered, the patient’s blood pressure suddenly decreased to about 60/40 mmHg and her heart rate was 70 beats/min. Massive bleeding was found in the surgical field. Her Hb level was 6.8 mg/dl at that time. The surgeon diagnosed the degree of placental adhesion as percreta, which had invaded the bladder and surrounding tissues. The bleeding was so extensive that the surgeon decided to perform a hysterectomy. Because of profound hypotension with a reduction in consciousness, the anesthetic method was changed to general anesthesia. Tracheal intubation was performed to secure the airway with rapid sequence intubation. An additional intravenous line was secured at the right external jugular vein, and rapid volume infusions and transfusions were performed using a rapid blood infuser (FMS 2000® fluid management system, Belmont Instrument Corp., USA). Despite attempts to ligate the uterine arteries, bleeding obscured the surgical field and resulted in further hemodynamic instability. The surgeon wished to perform a uterine artery embolization for bleeding control but it was difficult to maintain stability of the vital signs while transferring the patient to the intervention room. We, therefore, decided to implement a REBOA in the operation room to prevent hypovolemic shock. The acute care surgeon accessed the left femoral artery for the REBOA using a 7-French sheath (standard kit, Terumo, Vietnam) and inserted a balloon catheter (occlusion balloon catheter, Tokai Medical Products, Inc., Japan). After measuring the distance to the navel, 25 cm in this patient, they slid the balloon tip up to 25 cm from the punctured site and placed the balloon approximately between the infrarenal aorta and the bifurcation of the aorta. It was difficult to obtain radiologic confirmation of the balloon position because of the patient’s unstable vital signs. After balloon occlusion was accomplished, the systolic blood pressure immediately improved from 65 mmHg to 100 mmHg and the surgical field also improved (Fig. 2). REBOA was intermittently performed during the surgery and the balloon was sometimes partially deflated to identify the focus of the bleeding. During complete and partial inflation, ligation of both the uterine artery and internal iliac artery were performed. And the bleeding control was in progress by removing surrounding tissues where the placenta was invaded. The total duration of the occlusion was 75 min (divided over several occlusion times). The total operative time was 263 min and the total anesthesia time was 290 min. The estimated amount of blood volume lost was over 20,000 ml. Massive transfusions, including 36 units of packed red blood cells, 26 units of fresh-frozen plasma, and 10 units of platelets were administered.
Upon admission to the intensive care unit after embolization of the branch of the internal iliac artery, the patient was hemodynamically stable without any inotropes or vasopressors. No further transfusions were performed after surgery. The patient’s Hb was 12.2 g/dl, platelets were 89,000/μl, and prothrombin time (PT, international normalized ratio) and activated partial thromboplastin time (aPTT) were 13.9 s (1.23) and 39.1 s, respectively. The patient was extubated the same day and the femoral artery sheath was removed the next day. At postoperative day two, she was transferred to the ward and subsequently discharged on day 15 without complications. Neither the mother nor the baby experienced any complications after discharge.
DISCUSSION
PPH remains a major cause of maternal mortality and morbidity, so immediate awareness and quick and active efforts are needed to reduce poor outcomes [11]. This case demonstrated the successful resuscitation of a patient with life-threatening massive hemorrhage due to a MAP using REBOA. In our case, bleeding control was difficult and uterine artery embolization could not be performed immediately in the operating theater, so the REBOA helped maintain the blood pressure and save time transferring to the intervention room for further treatment.
Massive bleeding is a challenge to all involved medical staff, especially to the anesthesiologist who is monitoring the patient’s vital signs. Sometimes we anesthesiologists are faced with more bleeding than expected. Our patient had some risk factors associated with massive bleeding, including older age, a history of previous cesarean sections, and multiple parities. Therefore, we conducted arterial blood pressure monitoring and secured an additional intravenous line for volume administration. Two units of packed red blood cells were prepared for immediate use for bleeding. In our case, the initial diagnosis of the patient’s placenta was placenta previa with possible accreta but percreta was confirmed visually in the operating field. Because the sensitivity of ultrasound to diagnose MAP ranges from 33% to 93% [5], it may, unfortunately, provide a false-negative result. That means more severe adhesive placenta types can be found during surgery and more severe bleeding can be encountered unexpectedly.
Anesthesia for cesarean sections in MAP patients remains controversial but there are no differences in neonatal outcomes between regional and general anesthesia. While many anesthesiologists believe that general anesthesia is mandatory for cesarean sections for MAPs, such views are changing and many cesarean sections are successfully performed under regional anesthesia. Surgeon and the patient in our case also preferred regional anesthesia. Considering the risk factors of the patient previously described, a past medical history of multiple previous cesarean sections may lead to adhesions, which can prolong the delivery times and result in poor neonatal outcomes. These factors influenced the choice of anesthesia.
The prophylactic placement of REBOA can be considered for patients with abnormal placentation before surgery. According to several studies and reports, planned cesarean sections with the prophylactic use of a REBOA resulted in lower overall blood loss, fewer transfusions [12], and better maternal hemorrhage-related outcomes [13–15]. Thus, it seems reasonable to apply a REBOA in advance to patients who may have a risk of massive bleeding.
In spite of the potential effectiveness of REBOA in MAP patients [12,13], this approach has not been routinely adopted. To establish a system for facilitating the use of REBOA, involvement, time, and effort are required from the multidisciplinary team, including trained acute care surgeons and expert obstetrical surgeons, as well as trained anesthesiologists, because of the technical skills required. Furthermore, complications due to the procedure itself, including direct trauma to the aorta, hypoperfusion of vital organs, ischemia-reperfusion injury, thromboembolism, and hemorrhage can also occur, so these potential risks should be considered. In addition, the expense of medical care can be a burden. Considering the cost-effectiveness and complications of the technique itself, applying it prophylactically as a routine procedure may be a difficult choice. Even if the routine use of REBOA is difficult due to these limitations, we consider it a meaningful temporary technique for patients with uncontrolled massive PPH during surgery, such as in our case.
As a principal medical staff member responsible for monitoring the patient’s vital signs during surgery, anesthesiologists should be actively involved in decision-making and the operative plan. When we are faced with the unexpected situation of uncontrolled bleeding with a MAP, we should be able to consider and propose REBOA.
In conclusion, REBOA can be used as a rescue maneuver to decrease bleeding and improve the condition of the surgical field during an emergency PPH. We used REBOA in a patient with life-threatening PPH and it improved the patient’s vital signs and the condition of the surgical field temporarily. The patient was then managed successfully without further complications. Therefore, even though there are several limitations to the procedure, we recommend considering REBOA when faced with uncontrolled bleeding due to severe MAP.
Notes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conceptualization: Sung Mi Ji, Min A Kwon. Data acquisition: Chaemin Cho, Gunhwa Choi. Supervision: Min A Kwon. Writing-original draft: Sung Mi Ji, Chaemin Cho. Writing-review & editing: Jaegyok Song, Jeong Heon Park, Seokkon Kim.

